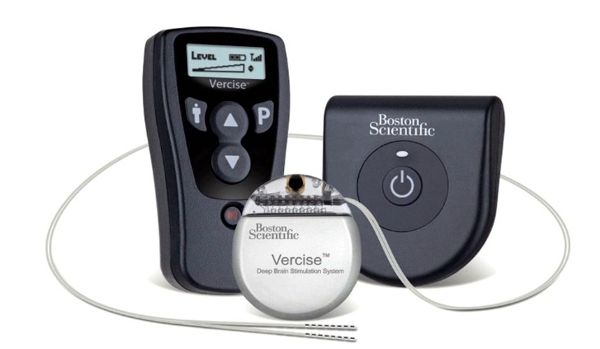
Boston Scientific announced today the first implantation of its Vercise™ DBS System as part of the INTREPID clinical trial. INTREPID is a prospective, multi-center, double-blinded, randomized, controlled study to evaluate the safety and effectiveness of motor function and overall quality of life in patients with the Vercise DBS System for the treatment of Parkinson’s disease. According to the press release:
Daily Archives: June 17, 2013
Implantabe Preterm Labor Monitor
 Not new, but recently came across while researching some information about related devices…
Not new, but recently came across while researching some information about related devices…
The NASA Tech Briefs of May 1999 described a device that telemeters pressure changes to detect intrauterine contractions. According to the brief:
A biotelemetric system for monitoring key physiological parameters of a fetus and its uterine environment is undergoing development. The main purpose of the monitoring is to detect preterm labor in order to enable timely treatment. At the present stage of development, the system monitors pressure changes and temperature. The pressure changes serve as direct indications of intrauterine contractions that could be associated with the onset of preterm labor. Future versions of the system are expected to monitor additional parameters, including pH and the heart rate of the fetus.