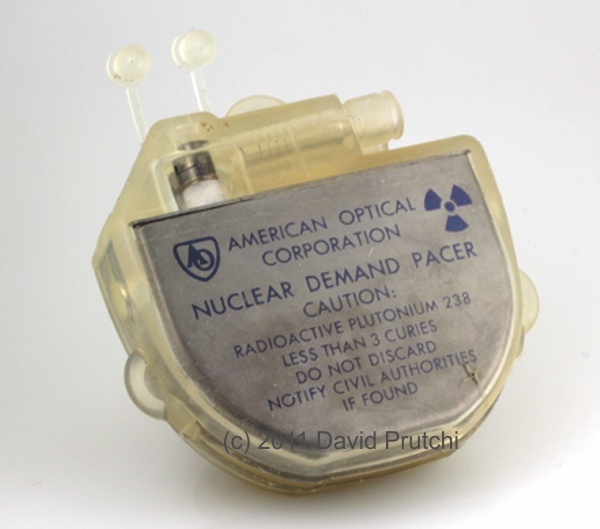
Active implantable medical devices are typically enclosed in a hermetically-sealed titanium housing which provides protection of the circuitry and other components.
Commonly, Grade 1 titanium is formed into the enclosure using stamping. The pretty, rounded shapes of modern pacemakers and ICDs are achieved by having two enclosure halves shallowly stamped from sheet stock material, and are then welded together to form the enclosure.
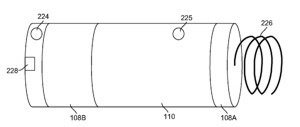
On the other hand, “boxy” enclosures that were popular in the 1970s are made by deeply drawing the titanium into a seamless can that is then capped by an ellipsoidal lid. However, many modern start-up companies still develop their early-generation implantable devices in this format because a number of off-the-shelf implantable-grade cells are available to fit in these deep-drawn enclosures, and the housings can be purchased off-the-shelf from various manufacturers. In addition, these enclosures provide sufficient volume for electronic assemblies manufactured using off-the-shelf packaged components and printed-circuit-board technology instead of more aggressive manufacturing methods (e.g. bare IC die on ceramic substrates) used by more established companies to miniaturize their mature implantable devices. Continue reading



 It has been known for quite some time that bone growth is stimulated by DC electrical currents. However, only relatively recently implantable stimulators have been developed for the application.
It has been known for quite some time that bone growth is stimulated by DC electrical currents. However, only relatively recently implantable stimulators have been developed for the application. BioControl Medical, Ltd. was founded in 1999 by
BioControl Medical, Ltd. was founded in 1999 by 
 Cameron Health was founded in 2000 in San Clemente, CA to develop a leadless implantable defibrillator.
Cameron Health was founded in 2000 in San Clemente, CA to develop a leadless implantable defibrillator.