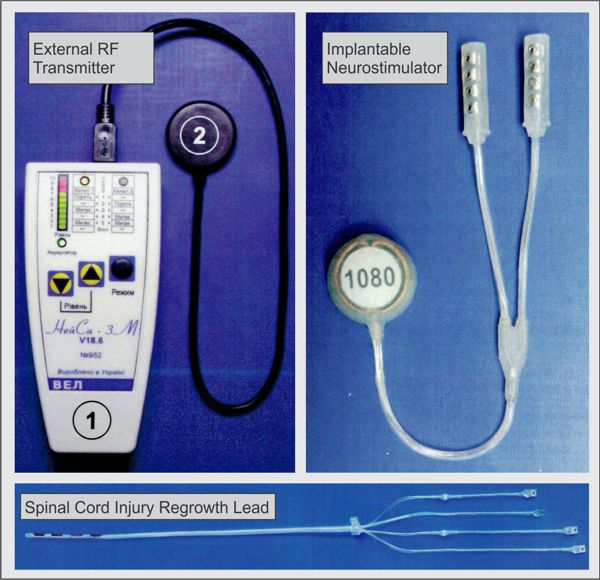
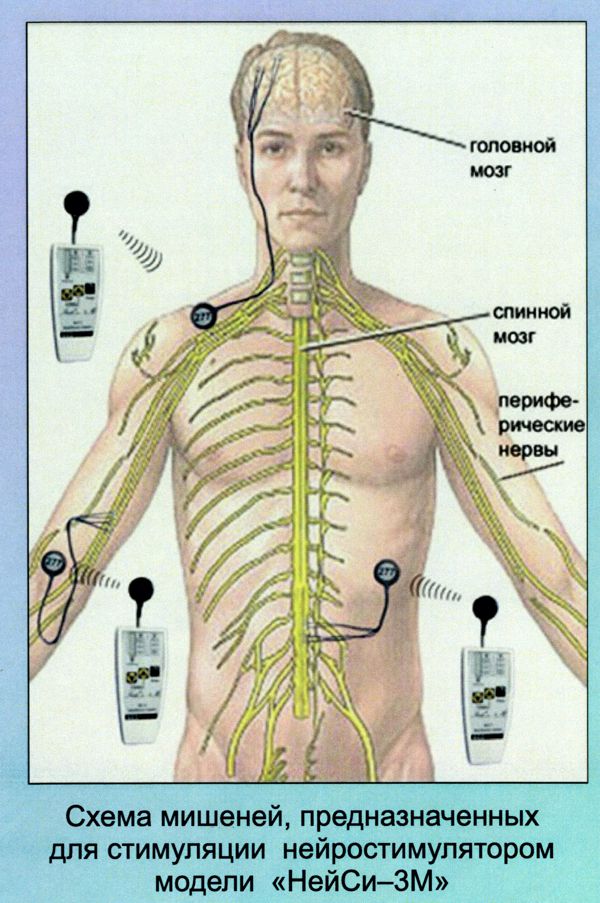
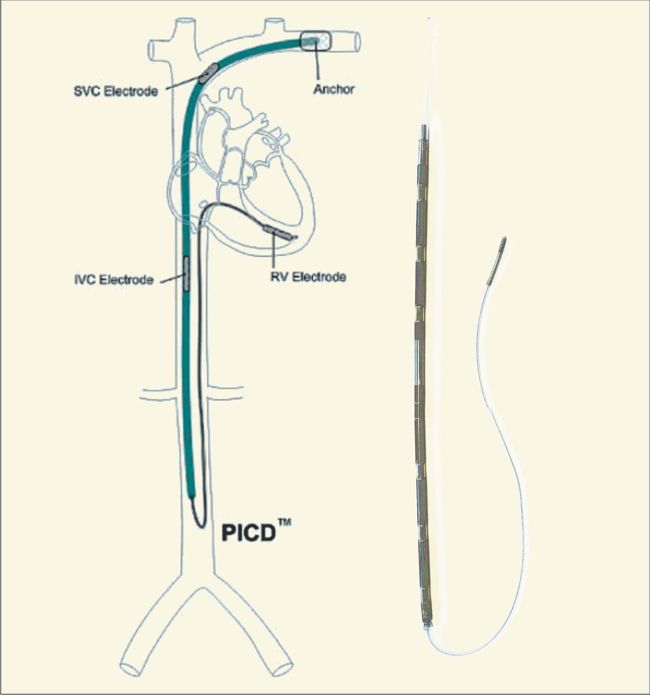
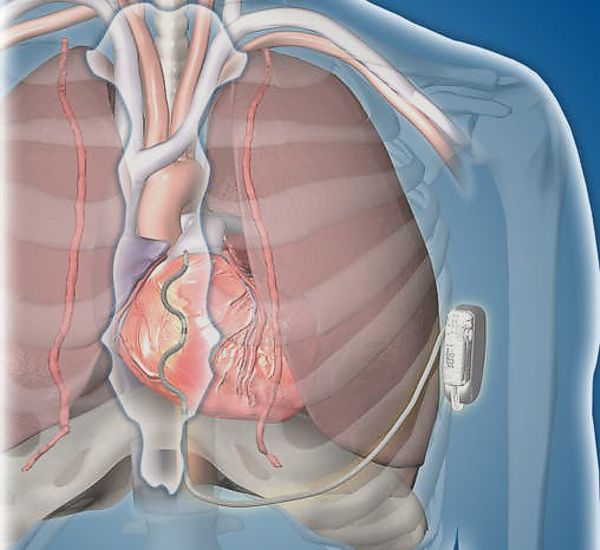
Image Credit: Medtronic
Medtronic announced today that it has received U.S. Food and Drug Administration (FDA) approval for the Attain Stability(TM) Quad MRI SureScan(TM) left heart lead. Paired with Medtronic quadripolar cardiac resynchronization therapy-defibrillators (CRT-D) and -pacemakers (CRT-P), the Attain Stability Quad lead is the only active-fixation left heart lead, and is designed for precise lead placement and stability. Medtronic said that the lead will be commercially available in the U.S. in summer 2019.

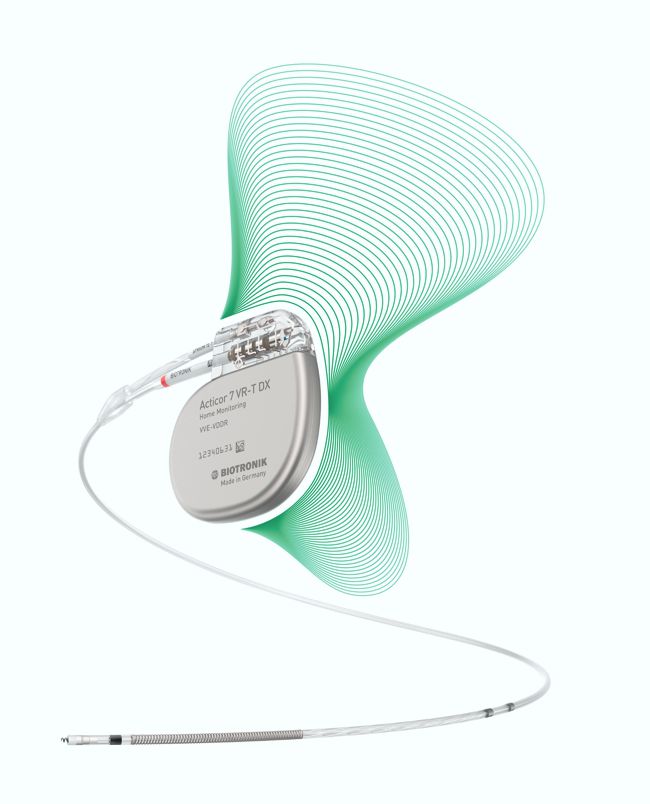





 I have led the development of devices to deliver CCM™ therapy from the very beginning, so it is with great pleasure and pride that I share with you the exciting news that Impulse Dynamics just received approval from the United States Food and Drug Administration (FDA) for our Optimizer® Smart System for heart failure patients! An official FDA announcement was made through the publication of a press release that you can find here:
I have led the development of devices to deliver CCM™ therapy from the very beginning, so it is with great pleasure and pride that I share with you the exciting news that Impulse Dynamics just received approval from the United States Food and Drug Administration (FDA) for our Optimizer® Smart System for heart failure patients! An official FDA announcement was made through the publication of a press release that you can find here: