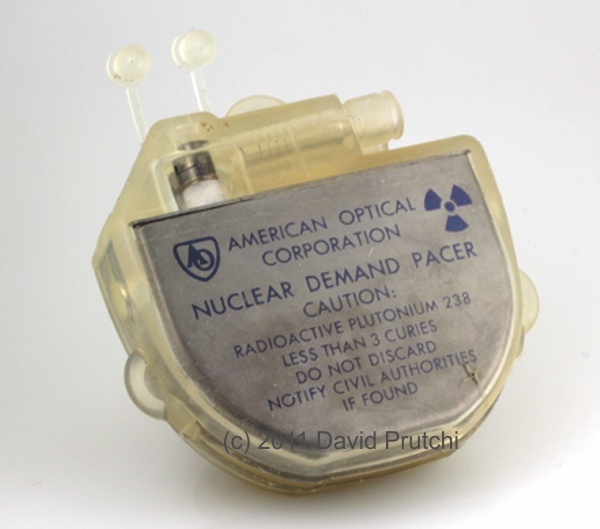
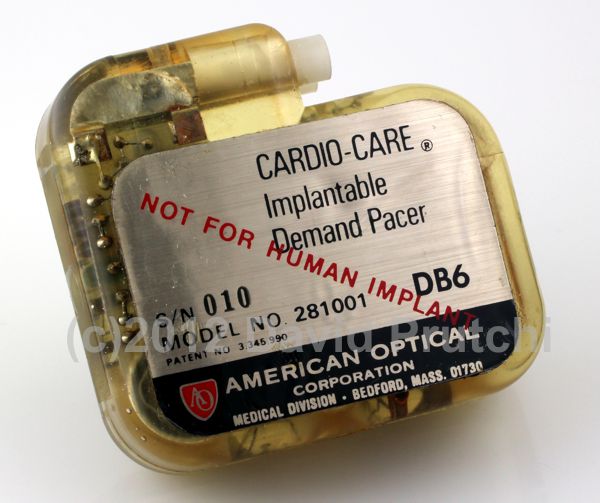
In 1973, former Medtronic sales representative Albert Beutel founded Intermedics in Freeport, TX. The first product was a small, mercury-cell-powered pacemaker. In 1974 Intermedics introduced a lithium-powered version, and in 1976 it introduced InterLith which was hermetically sealed, and weighed just 65 grams. At the time, InterLith’s size was a breakthrough, and became a very popular device, solidifying Intermedics’ position in the industry.







 One of the indicators of metabolic demand that has been used for controlling the rate of pacemakers is central venous blood temperature (CVT).
One of the indicators of metabolic demand that has been used for controlling the rate of pacemakers is central venous blood temperature (CVT).

 Leptos Biomedical was founded in Fridley, MN in 2002 by Dr. John D. Dobak. Leptos intended to develop an implantable device to stimulate the greater splanchnic nerve, that was hoped would result in reduced food intake and increased energy expenditure.
Leptos Biomedical was founded in Fridley, MN in 2002 by Dr. John D. Dobak. Leptos intended to develop an implantable device to stimulate the greater splanchnic nerve, that was hoped would result in reduced food intake and increased energy expenditure.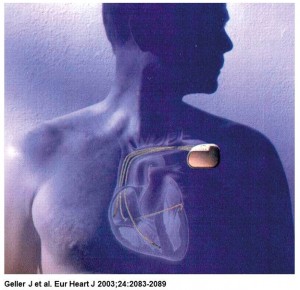 InControl was founded in 1990 in Redmond, WA to develop an implantable device for treating atrial fibrillation. In November 1995, InControl announced the first human implant of its Metrix atrioverter.
InControl was founded in 1990 in Redmond, WA to develop an implantable device for treating atrial fibrillation. In November 1995, InControl announced the first human implant of its Metrix atrioverter.