Mainstay Medical was founded in 2008 by entrepreneur Dr. Danny Sachs. The company’s ReActiv8 neurostimulator is intended to treat chronic low back pain caused by arthrogenic muscle inhibition. This is a neuroprotective condition in which the brain reflexively attempts to limit motor stimulation and movement in muscles surrounding a joint in response to damage or injury to the joint. In patients with this type of back pain, an initial injury triggers this reflexive inhibition, and the brain tries to limit painful movement by altering nerve transmissions to the deep stabilizing muscles of the spine, causing a loss of motor control, an unstable spine, and greater susceptibility to reinjury.
Category Archives: Neural Stimulation
AIMDs for the stimulation of the nervous system
Neuros Medical’s Altius™ High Frequency Electrical Nerve Block™ Pain Therapy
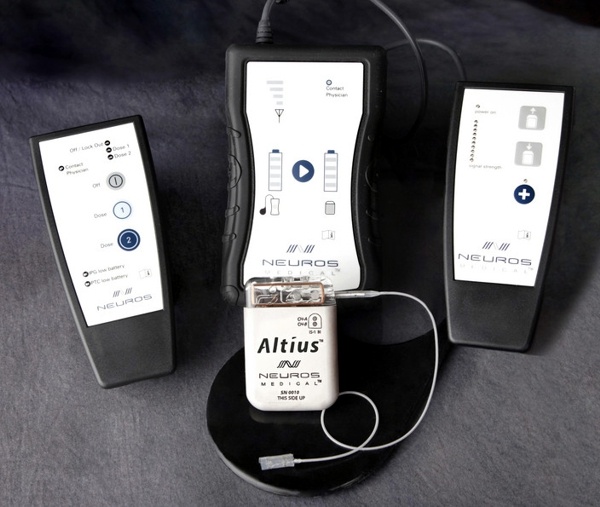
Neuros Medical, a Cleveland, Ohio based neuromodulation company, developed the Altius implantable system to deliver Electrical Nerve Block therapy for the treatment of chronic pain in a variety of applications including neuroma/residual limb pain, chronic post surgical pain, and chronic migraine.
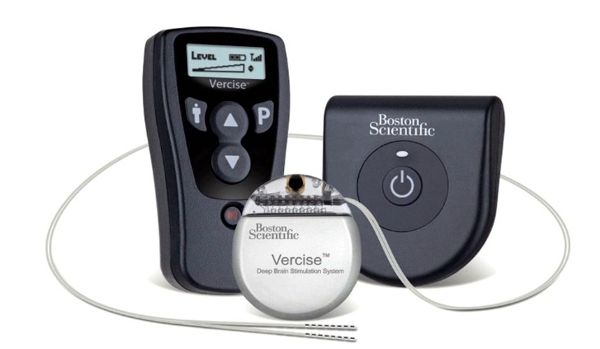
Boston Scientific Begins Patient Enrollment in U.S. Clinical Trial of Vercise™ DBS for Parkinson’s Disease, Presents EU Data
Boston Scientific announced today the first implantation of its Vercise™ DBS System as part of the INTREPID clinical trial. INTREPID is a prospective, multi-center, double-blinded, randomized, controlled study to evaluate the safety and effectiveness of motor function and overall quality of life in patients with the Vercise DBS System for the treatment of Parkinson’s disease. According to the press release:
Neurotech’s ADNS-300 Rechargeable Vagus Nerve Stimulator IPG for Epilepsy
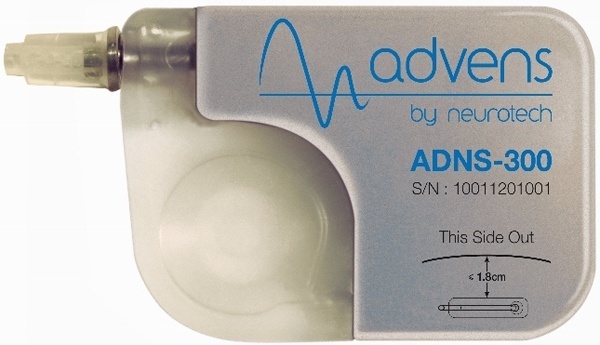
In June 2012 Neurotech s.a. (http://www.neurotech-int.eu), a Belgium based developer and manufacturer of neurostimulation devices, obtained the CE Mark for its ADNS-300 system for the treatment of refractory epilepsy. According to the press release:
“Advens therapy” uses the ADNS-300 stimulator as a therapeutic option for those patients who are refractory to anti-epileptic drugs and are not eligible for epilepsy (brain) surgery. The ADNS-300 is the world’s first rechargeable neurostimulator for vagus nerve stimulation. The device includes an innovative electrode and a promising “sensing” feature that may prove to be a significant technological breakthrough in the control of response to the therapy. Patients use a remote controller and charger to turn the system on/off and to recharge the neurostimulator. “Rechargeable systems may prove to be more cost-effective, especially if surgical operations to replace batteries are avoided over time. This helps patients to maintain a more independent life, with a lower risk of potential complications associated with battery replacement surgery.” ‘The role of Rechargeable Systems in Neuromodulation Paul Eldridge et al.’
St. Jude Medical Signs $40M Equity Investment and $300M Option with Spinal Modulation
 St. Jude Medical and privately-held Spinal Modulation, Inc., today announced that they have entered into a series of agreements under which St. Jude Medical made a $40 million equity investment in Spinal Modulation, a company that has developed an innovative neuromodulation therapy that provides a new pain management option for patients with chronic, intractable pain.
St. Jude Medical and privately-held Spinal Modulation, Inc., today announced that they have entered into a series of agreements under which St. Jude Medical made a $40 million equity investment in Spinal Modulation, a company that has developed an innovative neuromodulation therapy that provides a new pain management option for patients with chronic, intractable pain.
According to the announcement, “the agreement provides St. Jude Medical with an exclusive option to distribute the Axium™ Neurostimulator System, developed and manufactured by Spinal Modulation, in international markets where it is approved for sale. Additionally, St. Jude Medical will have the exclusive option to acquire the company for up to $300 million plus certain revenue-based milestones following U.S. commercialization.
Micromagnetic Stimulation as an Alternative to Electrical Stimulation for Implantable Devices? I don’t think so…

From: Microscopic magnetic stimulation of neural tissue, Giorgio Bonmassar, Seung Woo Lee, Daniel K. Freeman, Miloslav Polasek, Shelley I. Fried & John T. Gale, Nature Communications 3, Article number: 921
The following captured my attention in the announcement of the 11th World Congress of the International Neuromodulation Society, “Technology Transforming Chronic Illness Management.” From June 8 – 13, 2013:
“Micro-Magnetic Stimulation (Monday, June 10) – John T. Gale, Ph.D., has demonstrated for the first time that deep brain stimulation with micro-magnets can activate brain cells in a living organism. Dr. Gale’s research team has shown that placing a micro-magnet on the auditory pathway of hamsters triggers nerve signal transmission. Stimulation from uniquely designed magnetic fields could avoid unintentional activation of nearby brain areas and the associated side effects. Micro-magnets might one day provide stimulation for heart pacing, cochlear implants, Parkinson’s disease, or neural prosthetics.”
I have worked on TMS before, even home-brewed a TMS device (the design of which is detailed in my book “Design and Development of Medical Electronic Instrumentation: A Practical Perspective of the Design, Construction, and Test of Medical Devices”), but it takes a very large amount of energy to induce sufficient current in the tissue to stimulate excitable tissue, so it peaked my attention that to do so at the implantable level would be under consideration.
Boston Scientific Acquires and Launches Fixate Tissue Band for Spinal Cord Stimulator Leads and Pain Pump Catheters
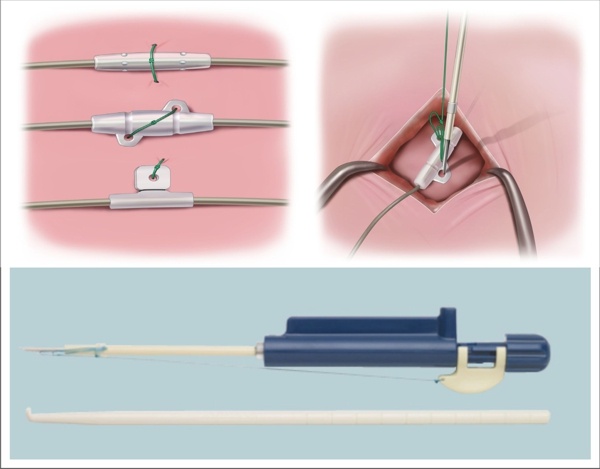
Boston Scientific announced today that it has acquired the fiXate™ Tissue Band and is launching it in the United States. The fiXate Tissue Band is intended to be an accessory to the leads/catheter component of Spinal Cord Stimulator/Pain Pump systems functioning to secure the lead to the fascia or inter-spinous/supra-spinous ligament. According to the press release:
“The fiXate Tissue Band is a novel suturing device that is designed to enable quick and simple placement of a suture to help secure a spinal cord stimulator (SCS) lead or pain pump catheter. Boston Scientific introduced the device earlier this month at the annual meeting of the American Academy of Pain Medicine in Fort Lauderdale. The product was purchased from Anulex Technologies, Inc.
Boston Scientific’s Precision Spectra™ 32-Contact SCS Launched in the US
 Boston Scientific received FDA approval and is beginning a limited launch of the Precision Spectra Spinal Cord Stimulator (SCS) System. According to the press release:
Boston Scientific received FDA approval and is beginning a limited launch of the Precision Spectra Spinal Cord Stimulator (SCS) System. According to the press release:
“The Precision Spectra System is the world’s first and only SCS system with Illumina 3D™ software and 32 contacts, and is designed to provide improved pain relief to a wide range of patients who suffer from chronic pain. Boston Scientific, the United States market leader in rechargeable SCS devices exiting 2012, is introducing the system at the annual meeting of the American Academy of Pain Medicine in Fort Lauderdale, Florida. Images of the Precision Spectra System can be downloaded here.
More than 100 million Americans suffer from chronic pain. Living in constant pain for an extended period of time can have a devastating impact on quality of life for many patients. Without relief, or the hope for relief, many patients lose the ability to sleep, work and function normally.
Medtronic’s Fully Implantable Percutaneous Nerve Stimulation (PNS) System for Treatment of Lower Back Pain
 In May 2011 Medtronic received the CE Mark for the first 16-electrode, fully implantable system for the percutaneous delivery of peripheral nerve stimulation (PNS) in the management of chronic back pain.
In May 2011 Medtronic received the CE Mark for the first 16-electrode, fully implantable system for the percutaneous delivery of peripheral nerve stimulation (PNS) in the management of chronic back pain.
PNS involves an implant of electrical leads just under the skin of the lower back. These leads are connected to a stimulator which delivers mild electrical impulses to the nerves, interrupting pain signals traveling through the nervous system to the brain.
Medtronic today announced the first patient enrollments in the SubQStim II pivotal clinical trial to pursue FDA approval PNS (also known as subcutaneous nerve stimulation or SQS), for the reduction of chronic, intractable post-surgical back pain.
St. Jude’s DBS Systems First to Receive CE Mark for Both Primary and Secondary Dystonia

St. Jude Medical announced CE Mark approval of its Brio™, Libra™ and LibraXP™ deep brain stimulation (DBS) systems for managing the symptoms of intractable primary and secondary dystonia, a neurological movement disorder that causes a person’s muscles to contract and involuntarily spasm, reducing the ability to control movement. This approval represents the first by a regulatory agency for the use of deep brain stimulation to manage both primary and secondary dystonia.
Retina Implant AG Publishes Study Results on its Alpha IMS Implant
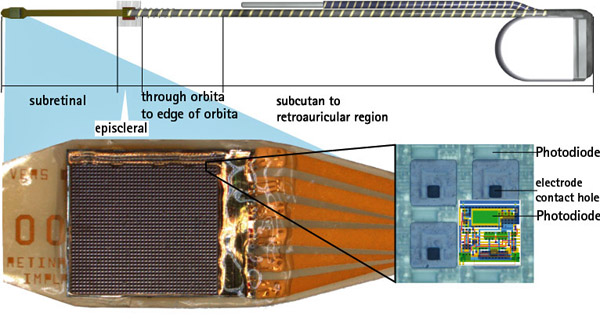
Retina Implant AG published study results on its Alpha IMS Implant for functional vision restoration. According to the press release, “Retina Implant AG, the leading developer of subretinal implants for patients blinded by retinitis pigmentosa (RP), announced results from part of its multicentre study were published today in the peer-reviewed journal Proceedings of the Royal Society B. The research found that, during the course of a three to nine month observation period, functional vision was restored in the majority of nine German patients implanted with a subretinal microchip as part of the first module of the Company’s second human clinical trial. In addition, visual acuity for two of the nine patients surpassed the visual resolution of patients from the Company’s first human clinical trial.
Second Sight Receives FDA Clearance for Argus II Retinal Prosthesis
On February 4, 2013, after more than 20 years of research and development, Second Sight Medical Products, Inc., announced that its Argus® II Retinal Prosthesis System (“Argus II”) has received U.S. market approval from the Food and Drug Administration (FDA) to treat individuals with late stage retinitis pigmentosa (RP). This announcement follows receipt of the European approval in 2011, and a unanimous recommendation by the FDA’s Ophthalmic Devices Advisory Panel in September 2012 that this revolutionary product be made available to treat this patient population in the U.S.
Medtronic Introduces First Neuromodulation Systems Compatible with Full-Body MRI
Medtronic has introduced in Europe the first and only implantable neurostimulation systems indicated for use in the treatment of chronic back and/or leg pain that are designed for full-body Magnetic Resonance Imaging (MRI) scans under specific conditions. Medtronic SureScan neurostimulation systems include enhancements to existing devices as well as specially designed leads to reduce or eliminate the hazards produced by the MRI environment. The devices also include a proprietary SureScan programming feature, which sets the device into an appropriate mode for the MRI environment.
SetPoint Medical’s Vagus Nerve Stimulation for Treatment of Systemic Inflammation
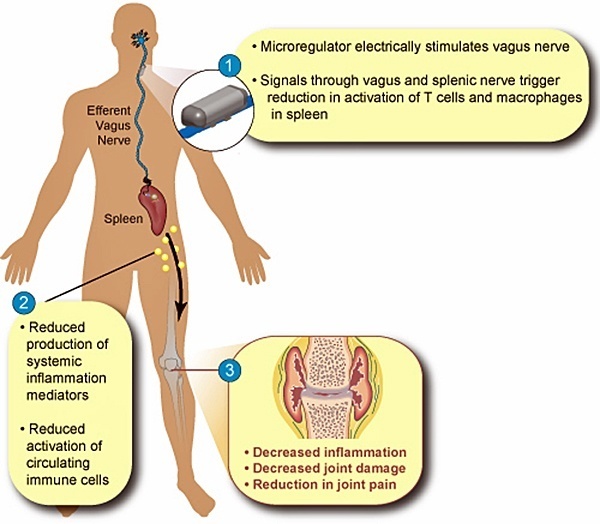
SetPoint Medical, headquartered in Valencia, California, is developing neuromodulation therapies for patients with inflammatory autoimmune diseases, such as rheumatoid arthritis (RA), inflammatory bowel disease (IBD), psoriasis, diabetes, heart disease, and multiple sclerosis. SetPoint’s proprietary neuromodulation platform consists of an implantable “microregulator”, wireless charger and iPad prescription pad application.
Functional Neuromodulation Ltd. Starts Study using DBS of the Fornix (DBS-f) for Mild Alzheimer’s
 Toronto-based Functional Neuromodulation announced that it implanted the first U.S. Alzheimer’s patient in the “ADvance Study” with a deep brain stimulation (DBS) system meant to improve cognitive performance. ADvance will evaluate the safety and potential clinical benefit of DBS of the fornix (DBS-f), a major inflow and output pathway in the brain’s memory circuit, for patients with mild Alzheimer’s. The ADvance Study is being conducted using Medtronic Activa DBS IPGs.
Toronto-based Functional Neuromodulation announced that it implanted the first U.S. Alzheimer’s patient in the “ADvance Study” with a deep brain stimulation (DBS) system meant to improve cognitive performance. ADvance will evaluate the safety and potential clinical benefit of DBS of the fornix (DBS-f), a major inflow and output pathway in the brain’s memory circuit, for patients with mild Alzheimer’s. The ADvance Study is being conducted using Medtronic Activa DBS IPGs.
While DBS has been an effective treatment for movement disorders for more than 15 years, it was only recently that this approach was first applied to Alzheimer’s. Dr. Andres Lozano, a neurosurgeon at University of Toronto and Scientific Founder of Functional Neuromodulation, originated the concept of treating memory disorders using deep brain stimulation (DBS) while treating a patient suffering from morbid obesity. In this patient, DBS stimulation of the hypothalamus and fornix was associated with an unexpected observed improvement in the patient’s memory.

![logo_mainstay1[1]](https://www.implantable-device.com/wp-content/uploads/2013/06/logo_mainstay11.jpg)