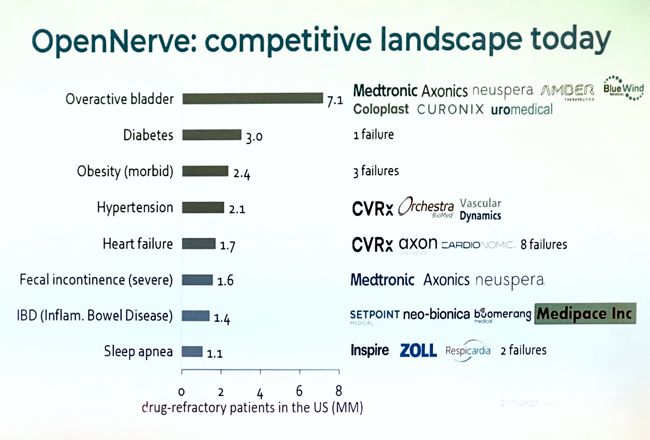
Image Credit: Medtronic
Medtronic announced that it received FDA’s approval for its Inceptiv™ closed-loop spinal cord stimulation system for the treatment of chronic pain.
According to the press release:
“Traditional fixed-output SCS devices deliver constant, mild electrical impulses that disrupt pain signals before they reach the brain. As patients go about their daily lives, certain movements such as laughing, bending, or sneezing may result in brief moments of uncomfortable overstimulation. This in turn may lead some patients to turn down their device’s stimulation output, resulting in a suboptimal therapy experience.
By contrast, Inceptiv SCS senses biological signals and consistently maintains the physician’s prescribed stimulation that is tailored to a patient’s needs. Specialized circuitry and a proprietary algorithm detect ECAPs (Evoked Compound Action Potentials), signals generated by the spinal cord in response to electrical stimuli. ECAPs are a direct measure of how much nerve tissue is activated in the spinal cord and can be used to inform real-time adjustments to stimulation. Inceptiv SCS senses the body’s response to stimulation 50 times per second and instantly increases or decreases stimulation to maintain prescribed settings as determined by the physician.”