(c) 2026 David Prutchi PhD
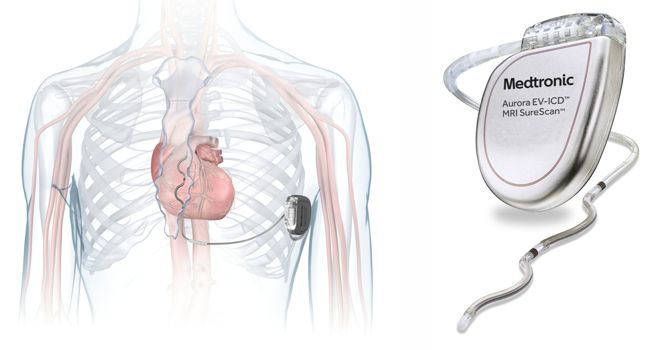
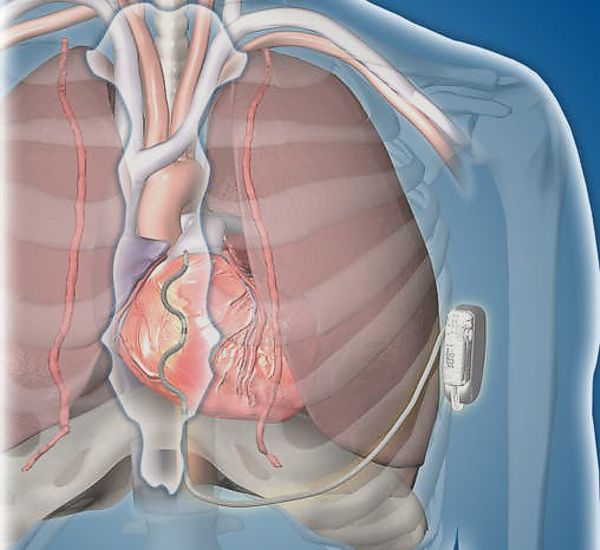
The Medtronic 5998 was a patient-activated implant used in the 1980s to manage tachyarrhythmias. The device was implanted to allow patients to trigger overdrive atrial pacing via a handheld transmitter to revert paroxysmal supraventricular tachycardia (PSVT).
The 5998R receiver was connected to a Medtronic 6991 right atrial lead. When the patient activated the 5998TDA transmitter, a burst of pacing was delivered to the heart. Brief periods (5 to 20 seconds) of rapid atrial pacing were effective in terminating the SVT and returning the heart to normal sinus rhythm. Documented issues with the device included inconsistent reversal of arrhythmias, and potential for transient atrial fibrillation.
The Medtronic 5998 RF system was in clinical use and described in the literature from at least 1981 through at least 1988.