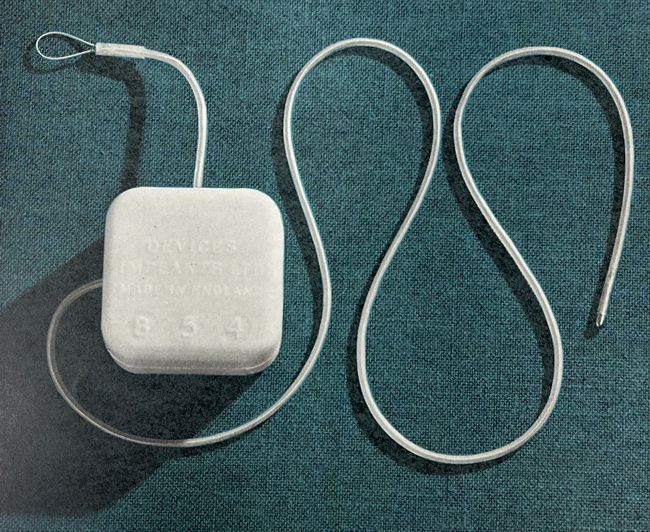
While curating HRS’ device collection, Dr. James Ong found this curious artifact. We tried looking for information about it online but came empty handed, so I decided to investigate further…
Here are my findings: Prutchi 2026 – General Electric A2074AA Pulse Sensor