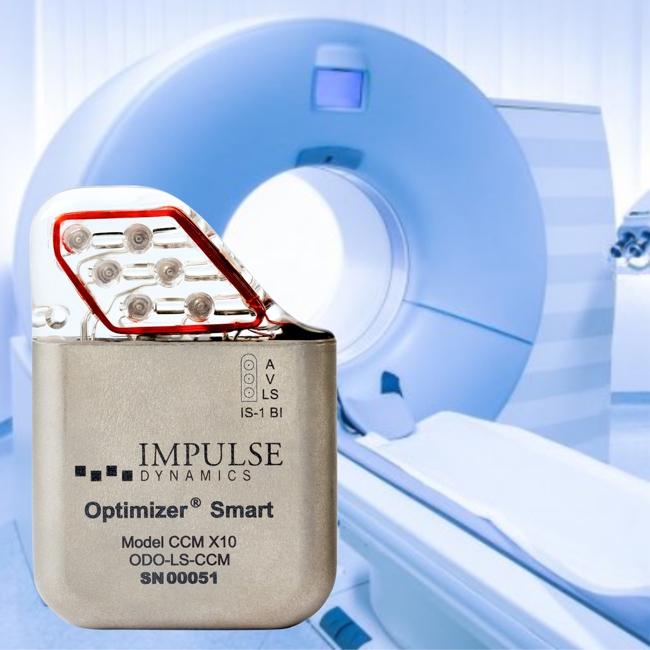
Impulse Dynamics – the company for which I am CTO and Executive VP – announced today that it received European approval for labeling the OPTIMIZER Smart IPG as MR-Compliant for full-body MRI scans utilizing 1.5 Tesla scanners:

Impulse Dynamics – the company for which I am CTO and Executive VP – announced today that it received European approval for labeling the OPTIMIZER Smart IPG as MR-Compliant for full-body MRI scans utilizing 1.5 Tesla scanners:

C-MIC Device. Image Credit: Berlin Heals
Berlin Heals just published the results of its first-in-human study of its C-MIC device, which was conducted in 10 NYHA Class III heart failure patients with LVEF<35%. The C-MIC is an implantable electroceutical device intended to readjust the disturbed myocardial electrical gradient that, according to Berlin Heals, occurs in heart failure patients.
In the study follow‐ups, a rapid and significant signal of efficacy (P < 0.005) was present with improvements in LVEF, left ventricular end‐diastolic diameter, left ventricular end‐systolic diameter, and distance walked. For eight patients, NYHA classification improved from Class III to Class I (for seven, as early as 14 days post‐operatively); for one, to Class II; and for one, to Class II/III. 36‐Item Short‐Form Health Survey questionnaire scores also improved highly significantly.
Berlin Heals has now initiated a randomized, controlled trial in 100 DCM patients, which will take place in Germany, Austria, Serbia, and Poland.
In addition, the FDA has granted Breakthrough Device designation for the C-MIC system
Berlin Heals website: http://berlinheals.de/
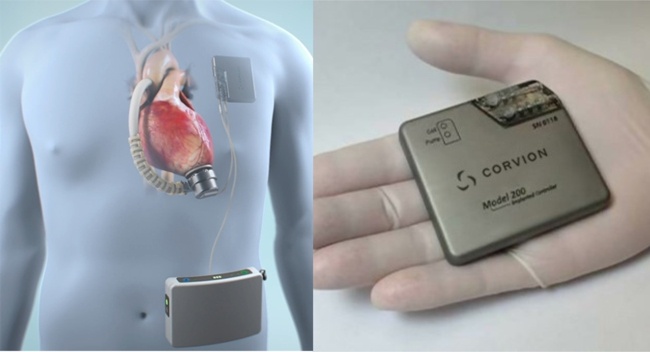
Image Credit: Corvion
Corvion is a pre-clinical stage company based in Webster, Texas. They are developing a fully-implantable LVAD that incorporates a highly efficient implantable rotary blood pump (Corvion claims 3X more efficient than competitors) coupled with a thin battery that gives 3 hours of free operation, and a robust transcutaneous energy transmission system.

Image credit: Onward
Onward is a company dedicated to the development of spinal cord stimulation therapies to improve function after spinal cord injury. Onward was formed in 2014 by researchers at the Swiss Federal Institute of Technology (EPFL). They are now headquartered at the High Tech Campus in Eindhoven, the Netherlands and the EPFL Innovation Park in Lausanne, Switzerland. Onward also has a growing team based in Boston, MA.
Medgadget carried today an interesting interview with Onward’s CEO regarding their technology (the ARC devices).
Onward’s website: onwd.com

Back in March of 2020, Second Sight Medical announced that it was winding down its operations. Second Sight developed implantable devices intended to create an artificial form of useful vision for blind individuals. However, because of the impact of COVID-19 on its ability to secure financing, it laid off the majority of its employees as a first step to an orderly wind-down of its operations.
The company had developed the Orion implant, which is placed directly onto the visual cortex of the patient’s brain. Signals received from a miniature camera integrated within a pair of glasses are fed to the implant and interpreted as “vision” by the brain. At the time of the closure announcement, the company was conducting a feasibility study with the Orion device implanted in six blind patients. Second Sight had been working towards a larger “pivotal” trial of the Orion implant, while all of the patients taking part in the small-scale study had reached the 12-month mark.
On January 6, 2021, Second Sight and France’s Pixium Vision announced that they have entered into a definitive business combination agreement pursuant to which Pixium Vision will, following the contribution to Second Sight of all of its assets and liabilities in relation to its neuromodulation technology used in the treatment of blindness, become the controlling shareholder of the new combined company, owning 60% of the total equity before the capital raise. The combined company will focus on retinal stimulation through Pixium’s Prima System. As part of the transaction, a new subsidiary will be created to focus on cortical stimulation through Orion. The new combined company will own 40% of the new subsidiary and will also have a first option to exclusive global marketing rights for Orion.

Pixium Vision’s Prima System. Image Credit: Images by Pixium Vision