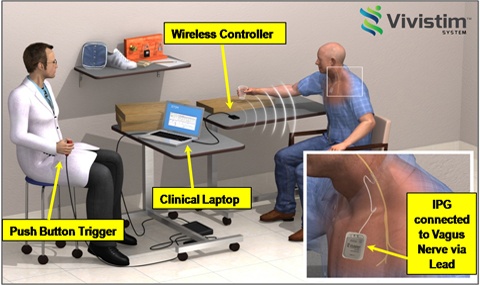
Image credit: MicroTransponder
MicroTransponder (now renamed Mobia Medical) developed the Vivistim™ system intended for the treatment of stroke. The implant is based on the idea that synchronizing vagus nerve stimulation with movement increases neural plasticity in the brain, resulting in enhanced recovery.
In May 2017 MicroTransponder presented results from its Vivistim™ Stroke Rehabilitation trial. The objective of this multicenter, double-blind, randomized, active-controlled trial was to improve upper limb mobility in stroke survivors suffering chronic disability. The data showed 75% of patients had a clinically meaningful response on the Fugl-Meyer Upper Extremity Scale (FMUE) at the end of a six weeks of physical therapy compared to 33% for the control group. 90 days after the six weeks of physical therapy ended 88% of VNS patients had responded compared to 33% in the control arm. And during the long-term portion of the study average FMUE scores continued to increase, with the 6 month and 9 month data showing further gains. All responders indicated they believed their overall health had improved and were satisfied with the therapy.
Over 1,000 Vivistim systems have been implanted so far.
In 2021 FDA approved the MicroTransponder Vivistim Paired VNS System to treat moderate to severe upper extremity motor deficits associated with chronic ischemic stroke using vagus nerve stimulation (VNS). Last year, Mobia reported sales of $32 million, which is approximately double of its prior-year. However, 2025 saw a net loss of $46.5 million, compared to a net loss of $24.6 million in 2024.
Recently, in February 2026, MicroTransponder announced that it has changed its corporate name to Mobia Medical, Inc. Now, Mobia has set the terms for its initial public offering, with the intent to raise about $150 million by offering 10 million shares for between $14 and $16 each. Mobia announced that it plans to use about $93.5 million of the IPO proceeds to expand its direct sales force and commercial organization. The proceeds of the IPO are expected to fund the company for around 1 year.