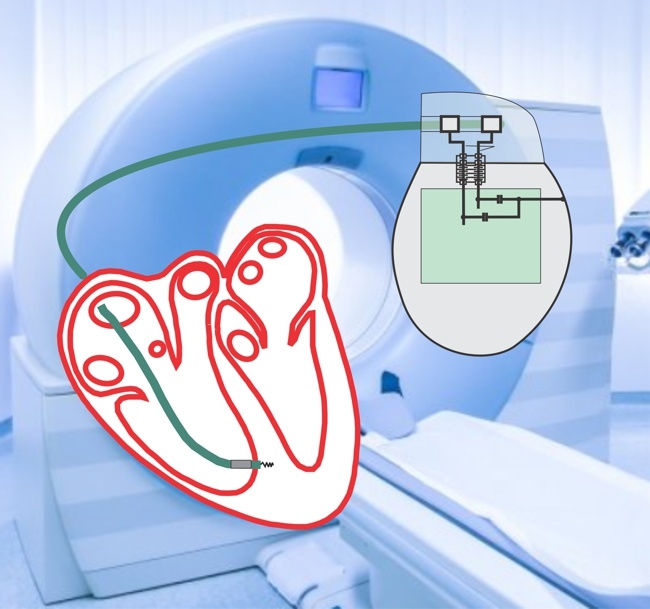
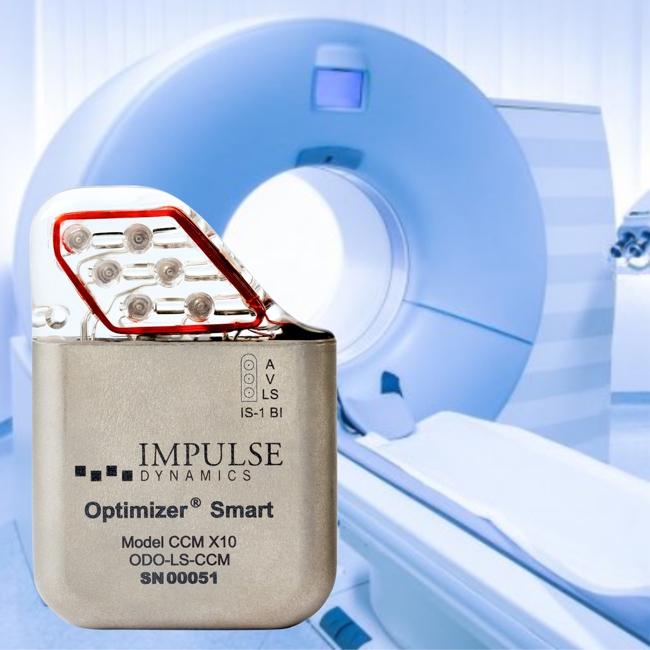
Image Credit: Impulse Dynamics
Impulse Dynamics, the company where I serve as Executive VP and CTO announced that the Centers for Medicare and Medicaid Services issued yesterday a favorable National Coverage Decision (NCD) memo for Cardiac Contractility Modulation (CCM®) Therapy. The decision was supported by the American College of Cardiology, the Heart Failure Society of America, and the Heart Rhythm Society.
According to the announcement:
“CCM® therapy for heart failure (HF) patients is one of only five therapies selected for the Transitional Coverage for Emerging Technologies (TCET) pathway in 2025. This determination marks a transformative milestone for HF patients and their providers, validating that CCM® therapy is no longer experimental or investigational.”




 Impulse Dynamics, the company where I’m CTO and Executive VP,
Impulse Dynamics, the company where I’m CTO and Executive VP,