
(c)2026 David Prutchi PhD. Photograph of device in the archive of the HRS.
Biophan Technologies was founded by Michael Weiner and Wilson Greatbatch in Pittsford, NY in December 2000 to develop solutions for making implantable devices compatible with MRI.
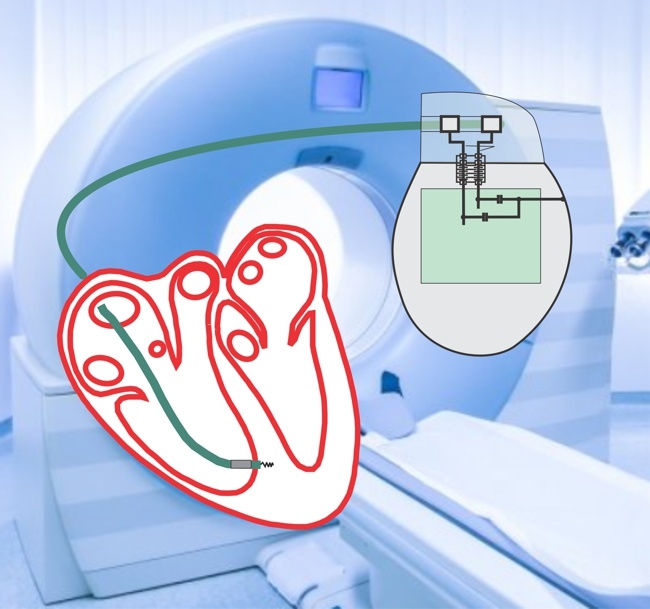
Since the vast majority of the MRI’s RF signal is picked up by the lead body, their concept was to have a laser pulse generator send light through an optical fiber lead body to illuminate a photovoltaic cell at the distal end to energize a bipolar electrode pair with the objective of making it MRI-safe.
The device shown in the picture is an external photonic pacemaker prototype in the HRS device archive. I don’t believe that an implantable version was ever built.
Biophan executed a license agreement with Boston Scientific Corporation in 2005, and sold its MRI safety patents to Medtronic for $11 million in 2007.








 Biotronik announced that it had received CE-approval for the world’s first DF4 ICD/CRT-D series approved for MRI. In addition, this series contains one of the world’s smallest ICDs– the Iforia single chamber ICD.
Biotronik announced that it had received CE-approval for the world’s first DF4 ICD/CRT-D series approved for MRI. In addition, this series contains one of the world’s smallest ICDs– the Iforia single chamber ICD.