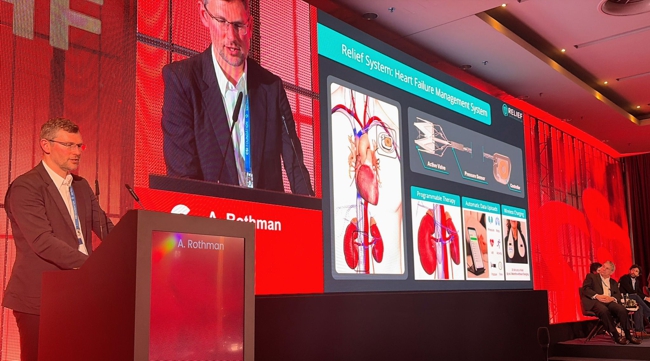
Render of what Relief Cardiovascular’s RELIEF System may look like. Based on image presented by Professor Alex Rothman at CSI congress’ Focus D-HF. Note: this is not an official Relief Cardiovascular image.
Relief Cardiovascular is a private early-stage company founded in 2022 and focused on transcatheter implants for heart-failure congestion, especially volume-overload and diuretic-resistant disease. It is backed by cardiovascular-focused investors including Broadview Ventures, Advent Life Sciences, Good Growth Capital, and Heartwork Capital.
The company’s lead product is the RELIEF System, which it describes as a permanently implanted transcatheter valve-and-sensor system intended to improve fluid management in heart failure by modulating inferior vena cava flow and monitoring venous hemodynamics. Relief says the implant is placed in the IVC below the renal veins, where it can reduce venous pressure, increase renal perfusion, and collect daily pressure waveforms for clinician-directed therapy through a cloud-enabled interface.
With this system, Relief is trying to move from temporary, hospital-based decongestion tools to a chronic implanted platform. Relief has said that temporary caval flow-modulating catheters have shown hemodynamic and clinical benefits but only provide temporary relief, whereas the Relief System is intended to provide autonomous hemodynamic monitoring together with the ability to modulate venous return over time.
Relief announced first-in-human procedures in September 2025. In March 2026, the company reported 90-day results from the prospective multicenter RELIEF-FIH study. According to their presentation at THT 2026, the study included 8 participants across 3 European sites and reported:
- 100% procedural success
- average implant procedure time of about 7 minutes from introducer insertion to removal
- zero device-related adverse events at 30 and 90 days
- hemodynamic measurement accuracy reported as R² = 0.9 versus right-heart catheterization
- sustained on-demand renal flow enhancement and pressure reduction with valve activation at 90 days.