Image credit: CCTV/The Chosun Daily
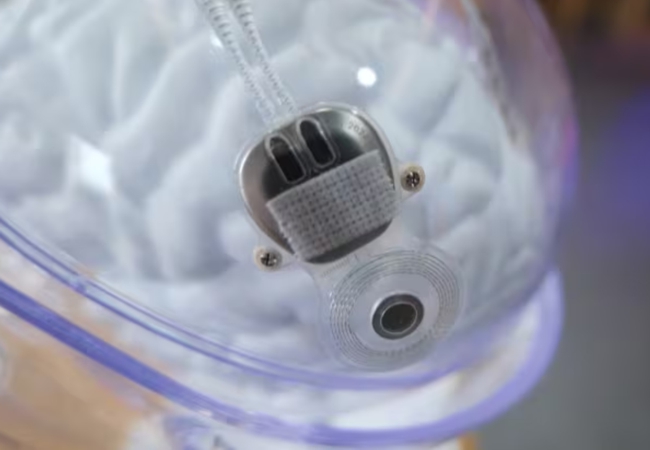
China’s National Medical Products Administration (CNMPA) recently approved an invasive BCI hand motor function compensation system developed by Shanghai-based BCI startup Neuracle.
Neuracle’s BCI (made by Borui Kang Medical Technology) is indicated for quadriplegic patients aged 16 to 60 who cannot use their hands due to cervical spinal cord injuries. The implant reads the patient’s extradural EEG and activates an exoskeletal glove in response to the patient’s thoughts.
The clinical trial supporting the approval showed that all 36 enrolled subjects could grasp specific objects simply by thinking. Most patients could control the device independently at home within a month post-surgery.
Other Chinese BCI companies nearing commercial approval include Shanghai-based NeuroXess and StairMed.
StairMed is pursuing the same invasive route as Neuralink, but has developed its own electrodes, algorithms, system hardware, and surgical robot. It is progressing through Chinese clinical work, including 64 channel and 256 channel systems.
NeuroXess, founded in 2021, and is focused on developing fully implantable, high-bandwidth flexible neural probes for medical applications.
