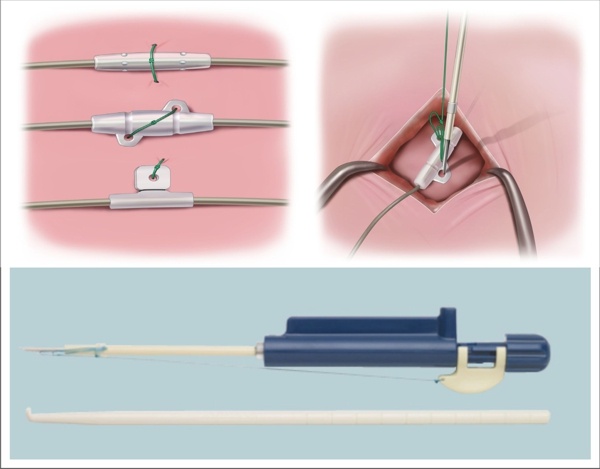
I have been honored with an invitation to present at the Fourth IEEE CASS Summer School on Wearable and Implantable Biomedical Circuits and Systems in Bogotá, Colombia (July 9 – 12, 2013).
I have been honored with an invitation to present at the Fourth IEEE CASS Summer School on Wearable and Implantable Biomedical Circuits and Systems in Bogotá, Colombia (July 9 – 12, 2013).
I will be giving two 1 hour and 20 minute talks on “A Practical Perspective on Developing Novel Commercial Active Implantable Medical Devices”. Unlike other commercial devices, developing medical implantable devices takes place in a heavily regulated environment which requires decisive proof of the devices’ safety and efficacy. Costs, schedules, and clinical strategies must be planned accordingly to achieve a successful exit. This two-part lecture will focus on the practical technical and business-oriented aspects of planning and executing the development of implantable medical devices intended for a commercial application.

 FDA has published a draft of the guidance document that it has developed to assist industry by identifying issues related to cybersecurity that manufacturers should consider in preparing premarket submissions for medical devices. This guidance document is intended to supplement FDA’s “Guidance for the Content of Premarket Submissions for Software Contained in Medical Devices” and “Guidance to Industry: Cybersecurity for Networked Medical Devices Containing Off-the-Shelf (OTS) Software”
FDA has published a draft of the guidance document that it has developed to assist industry by identifying issues related to cybersecurity that manufacturers should consider in preparing premarket submissions for medical devices. This guidance document is intended to supplement FDA’s “Guidance for the Content of Premarket Submissions for Software Contained in Medical Devices” and “Guidance to Industry: Cybersecurity for Networked Medical Devices Containing Off-the-Shelf (OTS) Software”

 St. Jude Medical and privately-held Spinal Modulation, Inc., today announced that they have entered into a series of agreements under which St. Jude Medical made a $40 million equity investment in Spinal Modulation, a company that has developed an innovative neuromodulation therapy that provides a new pain management option for patients with chronic, intractable pain.
St. Jude Medical and privately-held Spinal Modulation, Inc., today announced that they have entered into a series of agreements under which St. Jude Medical made a $40 million equity investment in Spinal Modulation, a company that has developed an innovative neuromodulation therapy that provides a new pain management option for patients with chronic, intractable pain.
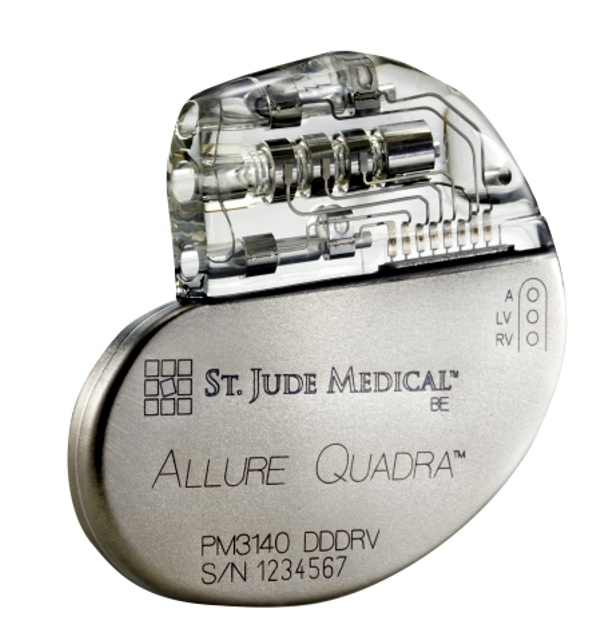 St. Jude Medical today announced CE Mark approval and European launch of its Allure Quadra™ Cardiac Resynchronization Therapy Pacemaker (CRT-P), which brings the quadripolar lead technology to the pacemaker market for the first time. According to the press release:
St. Jude Medical today announced CE Mark approval and European launch of its Allure Quadra™ Cardiac Resynchronization Therapy Pacemaker (CRT-P), which brings the quadripolar lead technology to the pacemaker market for the first time. According to the press release: