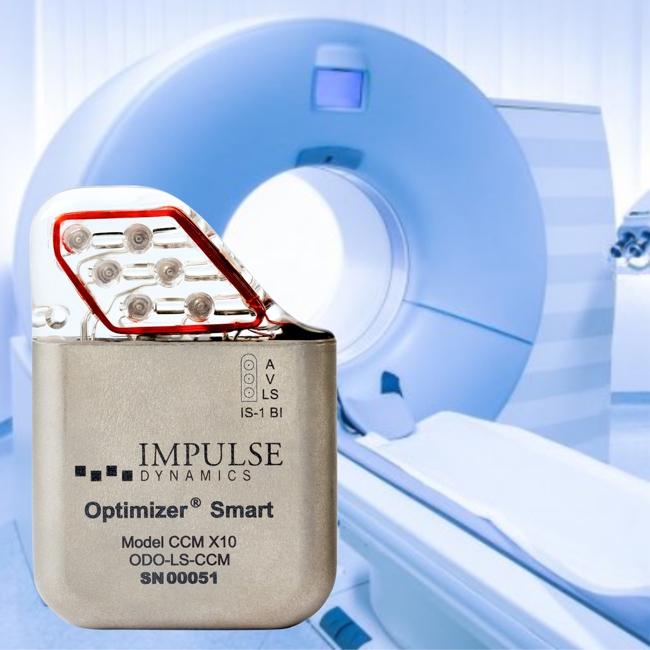
Impulse Dynamics – the company for which I am CTO and Executive VP – announced today that it received European approval for labeling the OPTIMIZER Smart IPG as MR-Compliant for full-body MRI scans utilizing 1.5 Tesla scanners:
Technologies for implantable devices

Impulse Dynamics – the company for which I am CTO and Executive VP – announced today that it received European approval for labeling the OPTIMIZER Smart IPG as MR-Compliant for full-body MRI scans utilizing 1.5 Tesla scanners:

Impulse Dynamics – the company for which I am CTO and Executive VP – received MR-conditional approval (1.5 T head/limbs with peripheral coils) from both FDA and the European Union.
From the press release:
IMPULSE DYNAMICS ANNOUNCES FDA APPROVAL FOR MAGNETIC RESONANCE IMAGING FDA
Clears Potential Hurdle for Many Heart Failure Patients
MARLTON, N.J.–(BUSINESS WIRE)–Impulse Dynamics, a company dedicated to improving the lives of people with heart failure (HF), today announced the U.S. Food and Drug Administration (FDA) has approved the conditional use of Magnetic Resonance Imaging (MRI) for Optimizer® CCM® delivery systems. This approval represents a significant advance because the population that benefits most from cardiac contractility modulation therapy (patients with moderate to severe HF) often requires advanced diagnostic imaging procedures.

Image Credit: iota Biosciences
iota Biosciences was established in 2017, building up on the concept of “neural dust” technology invented at the University of California, Berkeley by iota co-founders and co-CEOs Jose Carmena, Ph.D. and Michel Maharbiz, Ph.D.
iota’s “neural dust” consists of a small implantable device (a few mm long) that is powered from an external ultrasound generator. According to iota, their devices can record electrical information, stimulate nerves and communicate with other machines through ultrasound. Iota claims that “neural dust” devices can modulate the information transmitted through nerves, enabling doctors to better treat conditions from arthritis to cardiovascular disease
In 2018 iota completed a $15 million series A funding round aimed at developing a sensing platform. In September 2019, iota entered into a joint R&D agreement with Japanese Astellas Pharma to “design detailed specifications of implantable medical devices and conduct preclinical studies for several diseases with high unmet medical needs.”
Astellas today announced that it would acquire Iota in a deal that includes an initial payment of about $127.5M, covering the equity that Astellas did not acquire in 2018. An additional $176.5M are development-related milestone payments. Iota will be an independent subsidiary under Astellas’ U.S. umbrella.
Eventually, iota hopes to shrink the device to the size of grain of sand that can simultaneously sense neural activity and stimulate nerves to enable highly-targeted closed-loop therapies.
iota Biosciences’ website is at: https://iota.bio/
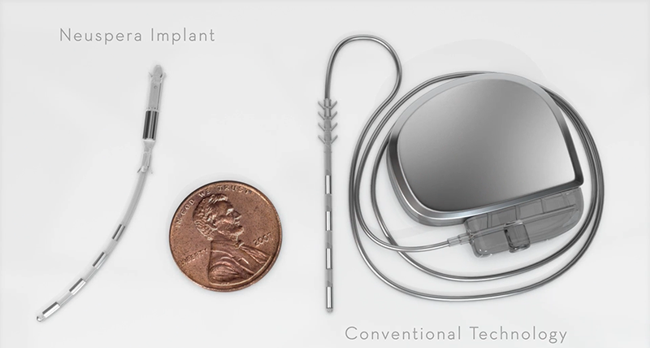
Image Credit: Neuspera Medical
Neuspera is a startup company located in San Jose, CA. They are developing miniaturized neuromodulation implants that are externally powered from a wearable device.
According to Neuspera, their “Mid-Field Powering” technology uses evanescent and propagating electromagnetic waves to power implanted medical devices to beyond 10cm of depth. Their technology is claimed to use the body as a natural waveguide to focus power ensuring energy is delivered to where it is needed.
In December 2019, Neuspera announced that it had received FDA’s approval to implant their systems under IDE. Neuspera now announced that it had performed the first human implants as part of their SANS-UUI two-stage seamless pivotal clinical trial to support FDA approval for patients with Urinary Urgency Incontinence (UUI), a symptom of overactive bladder.
Neuspera’s website: http://neuspera.com

Image Credit: Neuspera Medical

Image Credit: NeuroPace
NeuroPace of Mountain View, CA received FDA approval for MR-Conditional labeling of its RNS® implantable system for the treatment of medically refractory partial epilepsy.
Unlike Cyberonics’ VNS IPGs, the RNS® neurostimulator is designed to detect abnormal electrical activity in the brain and respond by delivering electrical stimulation to normalize brain activity before the patient experiences seizure symptoms. The neurostimulator is implanted in the cranium and connected to one or two leads that are implanted near the patient’s seizure focus.
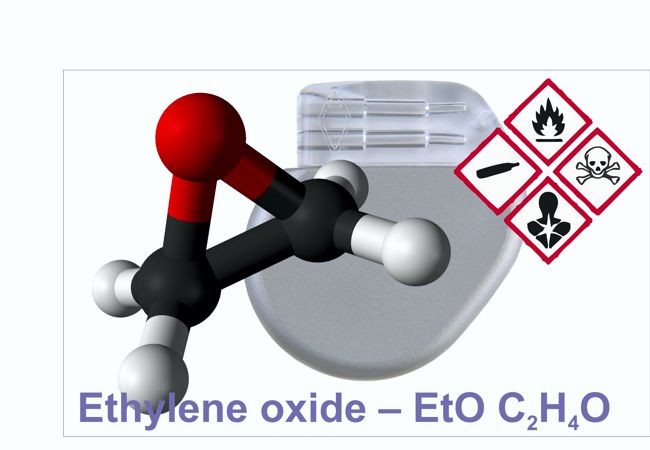
Ethylene oxide (EtO) is a chemical that is used to sterilize more than 50% of all medical device types and is crucial for preventing infection in patients undergoing surgeries and other medical treatments. For active implantable medical devices, EtO is pretty much the only option for sterilization. Alternative methods such as steam, radiation, or other sterilants either damage the device or do not achieve the needed levels of sterility assurance.
The US Environmental Protection Agency has been waging a war against the use of EtO by establishing an unreasonably low value for EtO concentration in its Integrated Risk Information System (IRIS). AdvaMed (the Advanced Medical Device Association) has urged the EPA to reassess its EtO value for one that is more feasible, based on the best available science and that will not potentially endanger the public health by threatening the availability of needed medical technologies.
AdvaMed President and CEO Scott Whitaker stated:
“… EPA’s EtO risk assessment standard is unworkable and not based on the best available science. The agency’s failure to address these valid scientific concerns surrounding their value threatens not only the medical technology supply chain but the tens of millions of American patients that rely on EtO-sterilized devices. We ask the agency to follow its own scientific recommendations and develop a revised EtO risk assessment standard that will effectively protect the public health and not disrupt patient access to needed medical technology.”
Click here to read AdvaMed’s complete response to the EPA.
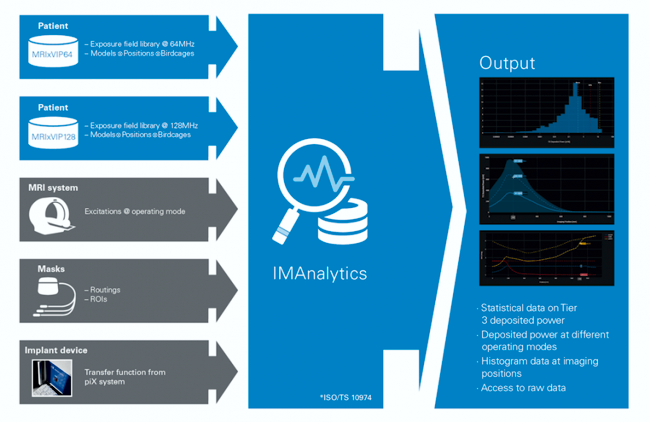
Image Credit: Zurich MedTech
The FDA announced the acceptance of Zurich MedTech’s Sim4Life IMAnalytics and the field exposure libraries MRIxVIP1.5T and MRIxViP3.0T from the IT’IS Foundation as a Medical Device Development tool (MDDT). This is the first FDA-approved computational modeling Medical Device Development Tool.
IMAnalytics is a software platform for the safety evaluation of implantable devices in the Magnetic Resonance (MR) environment. It characterizes RF-induced heating at the distal electrodes of implantable devices, using a variant of the Tier 3 approach as defined in ISO/TS 10974. It is tailored for elongated lead structures by making use of the transfer function method described in Annex K of the same technical specification.

Image Credit: Synchron
Synchron Inc. announced today the first successful clinical implantation of the Stentrode®, a minimally-invasive neural interface technology, a component of the Synchron Brain-Computer Interface. This is the first clinical feasibility trial evaluating this technology for its potential to restore communication in people with severe paralysis.

Image Credit: iota Biosciences
iota Biosciences was established in 2017, building up on the concept of “neural dust” technology invented at the University of California, Berkeley by iota co-founders and co-CEOs Jose Carmena, Ph.D. and Michel Maharbiz, Ph.D.
iota’s “neural dust” consists of a small implantable device (a few mm long) that is powered from an external ultrasound generator. According to iota, their devices can record electrical information, stimulate nerves and communicate with other machines through ultrasound. Iota claims that “neural dust” devices can modulate the information transmitted through nerves, enabling doctors to better treat conditions from arthritis to cardiovascular disease
In 2018 iota completed a $15 million series A funding round aimed at developing a sensing platform. In September 2019, iota entered into a joint R&D agreement with Japanese Astellas Pharma to “design detailed specifications of implantable medical devices and conduct preclinical studies for several diseases with high unmet medical needs.”
Eventually, iota hopes to shrink the device to the size of grain of sand that can simultaneously sense neural activity and stimulate nerves to enable highly-targeted closed-loop therapies.
iota Biosciences’ website is at: https://iota.bio/

I’ve stopped watching CNN, because it has become as bad and radically polarized as FOX news is on the other side of the spectrum, so it was an external reference that sent me to the following CNN article by Dominic Rech:
“Brain implants could give governments and companies power to read your mind, scientists warn“
So… now CNN is not only the mouthpiece for the far left, but like FOX News (which I consider a parody channel) has also turned into a pusher of ridiculous conspiracies.
Sure, neural implants can read specific motor patterns that, with exhaustive training of the subject, can be used as a brain-machine interface. However, that is an extremely far cry from being able to surreptitiously read someone’s mind or implant thoughts into people (AKA “synthetic telepathy”).
CNN’s Rech quotes a report by Dr. Tim Constandinou and his colleagues at the Next Generation Neural Interfaces (NGNI) Lab at Imperial College London. However, the report doesn’t imply anything of the sort of the sensationalistic title chosen by CNN.
Bottom line: NO, the Government and Private Companies will not be able to either read or place thoughts into your mind any time soon through brain implants!
Now, my main issue isn’t that CNN is using a ridiculous headline as click bait, but rather it is the fact that such reporting is irresponsible and outright dangerous.
Despite a clear notice that I won’t address such queries, I still receive copious amounts of e-mail messages from people who believe that they have been surreptitiously implanted with electronic devices to harass and spy on them. Mr. Rech’s article recklessly provides fuel for such psychotic thoughts, and legitimizes a conspiracy that discourages these people from receiving the psychiatric care they so critically require.
Shame on you Dominic Rech and CNN!

Image Credit: Boston Scientific
Boston Scientific announced the U.S. Food and Drug Administration (FDA) approval of its ImageReady™ MRI labeling for the Vercise Gevia™ Deep Brain Stimulation (DBS) System to be used in a full-body MRI (1.5 Tesla MRI conditional when all conditions of use are met). This system, with the Vercise Cartesia™ Directional Lead, is designed to treat the symptoms of Parkinson’s Disease (PD) by delivering precisely targeted electrical stimulation in the brain to provide optimal symptom relief and better control of unwanted side effects.

Image Credit: Synchron
Synchron is a Silicon Valley company that spun out of the University of Melbourne, Australia in 2012. It was started by Thomas Oxley, MD, PhD to develop a new concept for a Brain-Computer Interface
Synchron’s Stentrode™ is an endovascular neural interface. It is essentially an electrode array shaped as an endovascular stent that can be implanted via the jugular vein and advanced into the brain to the motor cortex. Neural signals are detected by the electrodes on the Stentrode™ and sent to a processing and communications unit implanted subcutaneously in the chest, and then wirelessly to an external receiver. The idea is that the device can interpret signals from the brain for patients with paralysis to control a computer operating system and set of applications that interact with assistive technologies.
Synchron is currently preparing for pilot clinical trials of the Stentrode™ to evaluate the safety and efficacy of this breakthrough technology.
Synchron’s website is at: https://www.synchronmed.com
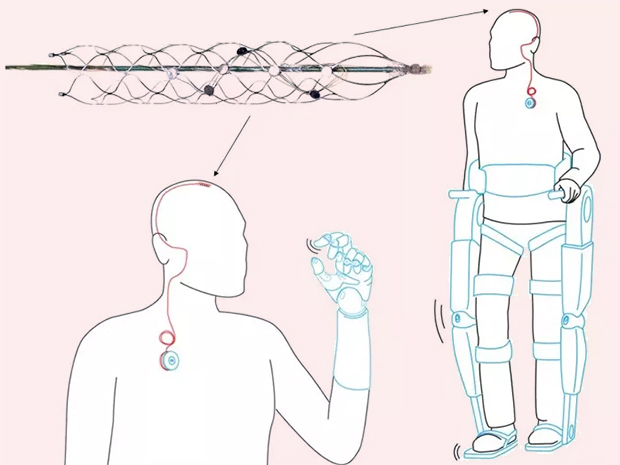
Conceptual use of the Stentrode device
Image Credit: University of Melbourne

Image Credit: Composite of images by Leviticus Cardio
Leviticus Cardio, a company based in Petach Tikva, Israel, has been developing a wireless power transfer technology that they call “Coplanar Energy Transfer” (CET). The system supports high-efficiency (up to 75%) power transfer levels of up to 30 Watts, making it suitable for powering ventricular-assist devices.
Like other transcutaneous energy transfer systems, Leviticus is used on inductive power transfer between an external primary coil and an implanted secondary coil. However, instead of the traditional pancake coil subcutaneous implant, the CET coil is placed around the lung. The unique engineering of the coplanar energy transfer system is characterized by two large rings utilizing a coil-within-the-coil topology, ensuring robust resonance energy transfer Continue reading→

Bioness is a Valencia, California medical device/rehabilitation company founded by the late Alfred Mann some 15 years ago. He had previously acquired a miniature stimulator called the BION, and a company called NESS who had a footdrop stimulator. The combination of the two is where the name Bioness came from.
One of its products is the “StimRouter”, which is an externally-powered implantable peripheral nerve stimulator designed to reduce chronic pain. It gained FDA approval in 2015 and was launched in mid-2016.
Unlike most externally-powered neurostimulators however, the StimRouter is not powered by RF received from the external transmitter. Instead, the external pulse transmitter is more like a TENS unit with gelled electrodes applied to the skin. The implant has no electronic components. It is just a lead that has a coil electrode that intercepts part of the current between the TENS electrodes, and routes the captured current to small electrodes in contact with the target nerve at the distal end of the lead.

Image Credit: Vectorius Medical
Vectorius Medical was founded in 2011 in the RadBioMed incubator in Tel-Aviv, Israel. It developed the V-LAP implantable device for the measurement of Left Atrial Filling Pressure (LAP).
The device is implanted in the left atrium, has no battery, and is able to communicate with an external interrogator from deep within the body using high-resolution waveform morphology. According to Vectorius Medical:
“Since the pressure of the heart’s left atrium is the earliest and most accurate real-time indication of heart failure exacerbation, the actionable feedback provided by the V-LAP will enable a significant improvement in ongoing management of heart failure patients. Once patients are implanted with the V-LAP, they will be able to measure left atrial pressure (LAP) daily at home via an easy, non-invasive method using a small, portable external unit.
…
Left atrial pressure has been scientifically established as the most specific physiological indication for heart failure exacerbation. Approximately 90% of patients admitted to the hospital for heart failure have pulmonary congestion related to elevated left atrial filling pressure (LAP). The increase of LAP is the earliest sign of impending heart failure exacerbation – long before clinical symptoms occur.”
The Vectorius system is intended to provide daily readings patient hemodynamic pressure to provide early, accurate physiological indication of cardiac decompensation. Physicians can access data through a Cloud-based system.
On February 4, 2019, Vectorius announced the initiation of the VECTOR-HF First-in-Human (FIH) clinical trial, and the successful first ‘in-human’ implantation of the V-LAP monitoring device. According to Vectorius Medical:
“Implantation of the device in this first “In-Human” trial was completed in just six minutes. It was fixated within the patient’s interatrial septum of the heart using a standard minimally-invasive percutaneous procedure under fluoroscopy and echocardiographic guidance, with the application of local anesthesia.”
Vectorius’ website is at: http://vectoriousmedtech.com