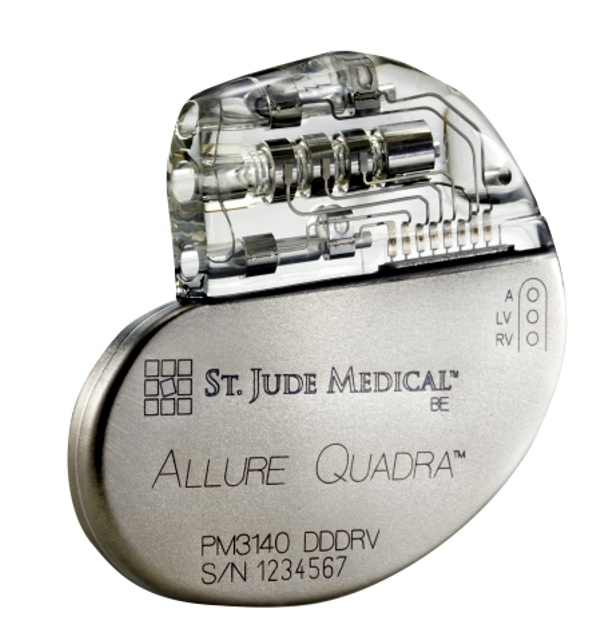
 St. Jude Medical today announced CE Mark approval and European launch of its Allure Quadra™ Cardiac Resynchronization Therapy Pacemaker (CRT-P), which brings the quadripolar lead technology to the pacemaker market for the first time. According to the press release:
St. Jude Medical today announced CE Mark approval and European launch of its Allure Quadra™ Cardiac Resynchronization Therapy Pacemaker (CRT-P), which brings the quadripolar lead technology to the pacemaker market for the first time. According to the press release:
“Quadripolar leads allow for increased implant efficiencies, which clinical data indicates can result in fewer surgical revisions. The Allure family of devices also offer enhanced heart failure (HF) diagnostics, including CorVue™ Impedance Monitoring, for improved patient management.


 Biotronik announced that it had received CE-approval for the world’s first DF4 ICD/CRT-D series approved for MRI. In addition, this series contains one of the world’s smallest ICDs– the Iforia single chamber ICD.
Biotronik announced that it had received CE-approval for the world’s first DF4 ICD/CRT-D series approved for MRI. In addition, this series contains one of the world’s smallest ICDs– the Iforia single chamber ICD.







 In 1965, Australian medical device pioneer Noel Gray established Telectronics – Australia’s first manufacturing facility for producing pacemakers that were designed in-house. Telectronics was an innovative developer, achieving some major successes in the early cardiac pacing field, for example, Telectronics’ leads allowed narrowing the pacing pulse to its current nominal of 0.5 milliseconds; encapsulating the pacemaker in titanium instead of epoxy; using a microplasma weld to join the two halves of the pacemaker capsule; creating one of the first rate-responsive ‘demand’ pacemakers; and isolating the pacemaker’s battery in a separate compartment to deal with the problem of leaking mercury-zinc batteries.
In 1965, Australian medical device pioneer Noel Gray established Telectronics – Australia’s first manufacturing facility for producing pacemakers that were designed in-house. Telectronics was an innovative developer, achieving some major successes in the early cardiac pacing field, for example, Telectronics’ leads allowed narrowing the pacing pulse to its current nominal of 0.5 milliseconds; encapsulating the pacemaker in titanium instead of epoxy; using a microplasma weld to join the two halves of the pacemaker capsule; creating one of the first rate-responsive ‘demand’ pacemakers; and isolating the pacemaker’s battery in a separate compartment to deal with the problem of leaking mercury-zinc batteries. 