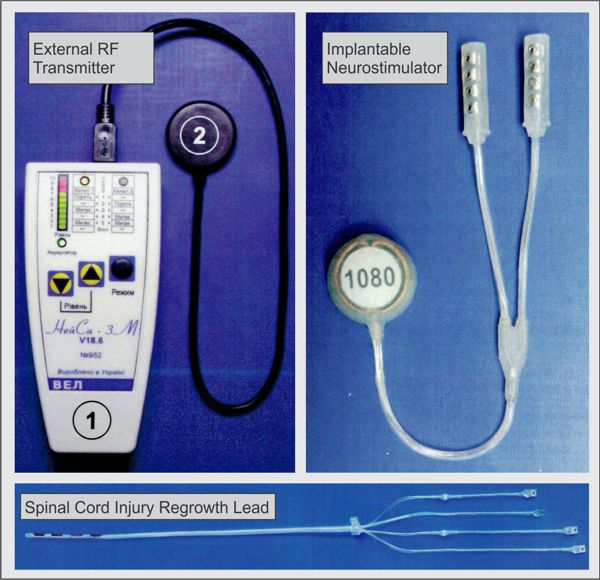
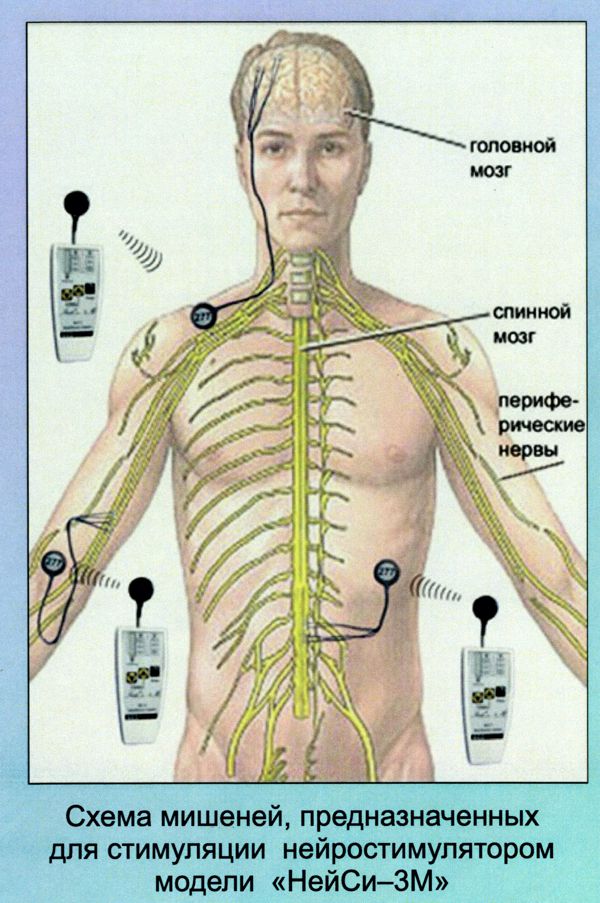
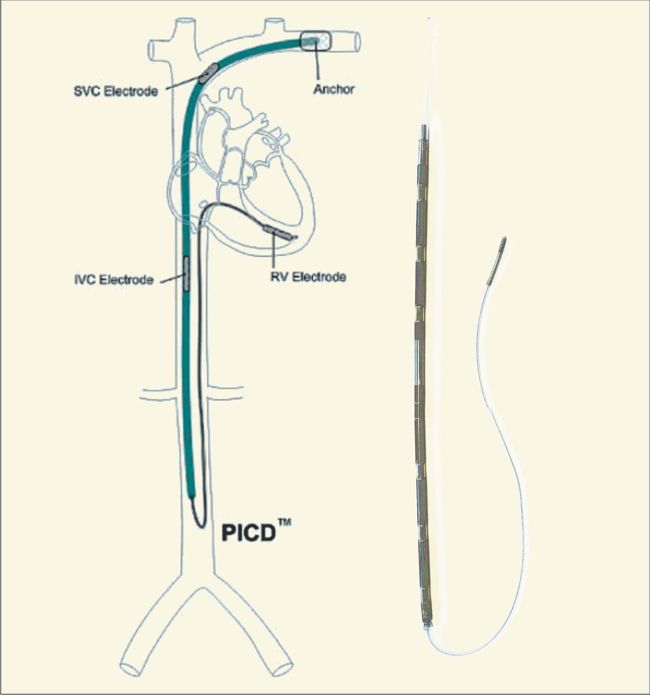
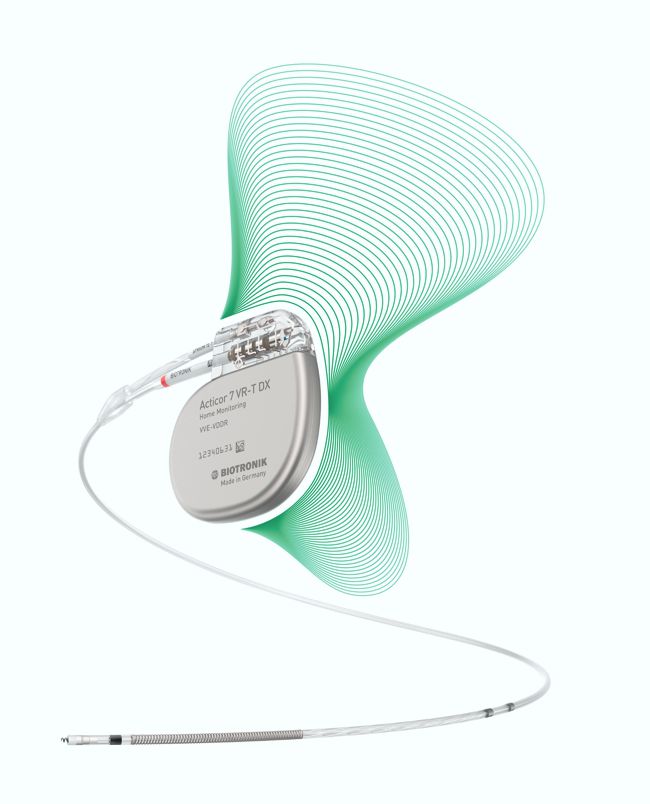
 Biotronik announced yesterday the full commercial launch of the Acticor device family, including Acticor DX and CRT-DX devices. The unique feature of the system is the single-pass lead with its floating atrial sensing rings. According to the press release:
Biotronik announced yesterday the full commercial launch of the Acticor device family, including Acticor DX and CRT-DX devices. The unique feature of the system is the single-pass lead with its floating atrial sensing rings. According to the press release:
“When implanted with Biotronik’s Plexa ProMRI S DX lead, the hybrid ICD Acticor systems offer dual-chamber diagnostics without the need for an atrial lead. Importantly, all three Acticor DX devices feature a new DF4 header configuration with a penta-polar electrode lead cable design that simplifies the implant procedure for physicians.”





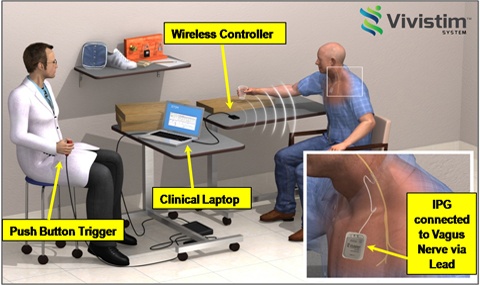
 I have led the development of devices to deliver CCM™ therapy from the very beginning, so it is with great pleasure and pride that I share with you the exciting news that Impulse Dynamics just received approval from the United States Food and Drug Administration (FDA) for our Optimizer® Smart System for heart failure patients! An official FDA announcement was made through the publication of a press release that you can find here:
I have led the development of devices to deliver CCM™ therapy from the very beginning, so it is with great pleasure and pride that I share with you the exciting news that Impulse Dynamics just received approval from the United States Food and Drug Administration (FDA) for our Optimizer® Smart System for heart failure patients! An official FDA announcement was made through the publication of a press release that you can find here: