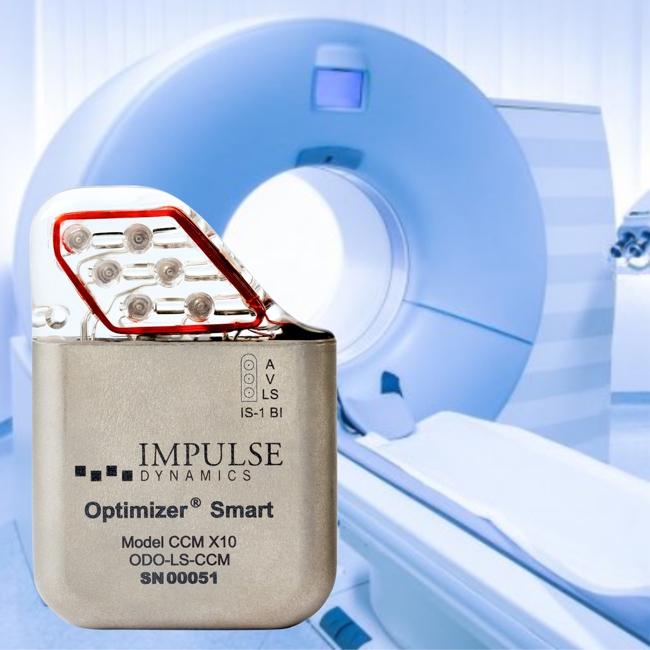
Impulse Dynamics – the company for which I am CTO and Executive VP – announced today that it received European approval for labeling the OPTIMIZER Smart IPG as MR-Compliant for full-body MRI scans utilizing 1.5 Tesla scanners:

Impulse Dynamics – the company for which I am CTO and Executive VP – announced today that it received European approval for labeling the OPTIMIZER Smart IPG as MR-Compliant for full-body MRI scans utilizing 1.5 Tesla scanners:

C-MIC Device. Image Credit: Berlin Heals
Berlin Heals just published the results of its first-in-human study of its C-MIC device, which was conducted in 10 NYHA Class III heart failure patients with LVEF<35%. The C-MIC is an implantable electroceutical device intended to readjust the disturbed myocardial electrical gradient that, according to Berlin Heals, occurs in heart failure patients.
In the study follow‐ups, a rapid and significant signal of efficacy (P < 0.005) was present with improvements in LVEF, left ventricular end‐diastolic diameter, left ventricular end‐systolic diameter, and distance walked. For eight patients, NYHA classification improved from Class III to Class I (for seven, as early as 14 days post‐operatively); for one, to Class II; and for one, to Class II/III. 36‐Item Short‐Form Health Survey questionnaire scores also improved highly significantly.
Berlin Heals has now initiated a randomized, controlled trial in 100 DCM patients, which will take place in Germany, Austria, Serbia, and Poland.
In addition, the FDA has granted Breakthrough Device designation for the C-MIC system
Berlin Heals website: http://berlinheals.de/
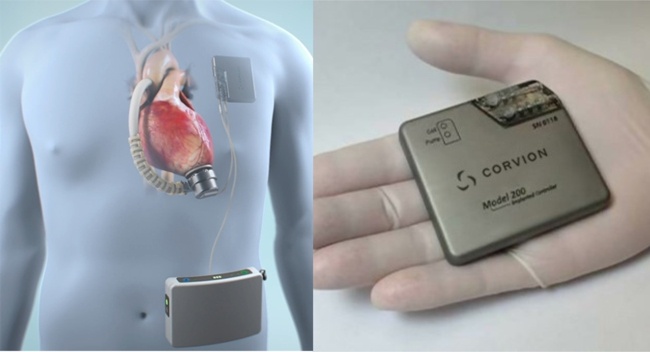
Image Credit: Corvion
Corvion is a pre-clinical stage company based in Webster, Texas. They are developing a fully-implantable LVAD that incorporates a highly efficient implantable rotary blood pump (Corvion claims 3X more efficient than competitors) coupled with a thin battery that gives 3 hours of free operation, and a robust transcutaneous energy transmission system.

Image credit: Onward
Onward is a company dedicated to the development of spinal cord stimulation therapies to improve function after spinal cord injury. Onward was formed in 2014 by researchers at the Swiss Federal Institute of Technology (EPFL). They are now headquartered at the High Tech Campus in Eindhoven, the Netherlands and the EPFL Innovation Park in Lausanne, Switzerland. Onward also has a growing team based in Boston, MA.
Medgadget carried today an interesting interview with Onward’s CEO regarding their technology (the ARC devices).
Onward’s website: onwd.com

Back in March of 2020, Second Sight Medical announced that it was winding down its operations. Second Sight developed implantable devices intended to create an artificial form of useful vision for blind individuals. However, because of the impact of COVID-19 on its ability to secure financing, it laid off the majority of its employees as a first step to an orderly wind-down of its operations.
The company had developed the Orion implant, which is placed directly onto the visual cortex of the patient’s brain. Signals received from a miniature camera integrated within a pair of glasses are fed to the implant and interpreted as “vision” by the brain. At the time of the closure announcement, the company was conducting a feasibility study with the Orion device implanted in six blind patients. Second Sight had been working towards a larger “pivotal” trial of the Orion implant, while all of the patients taking part in the small-scale study had reached the 12-month mark.
On January 6, 2021, Second Sight and France’s Pixium Vision announced that they have entered into a definitive business combination agreement pursuant to which Pixium Vision will, following the contribution to Second Sight of all of its assets and liabilities in relation to its neuromodulation technology used in the treatment of blindness, become the controlling shareholder of the new combined company, owning 60% of the total equity before the capital raise. The combined company will focus on retinal stimulation through Pixium’s Prima System. As part of the transaction, a new subsidiary will be created to focus on cortical stimulation through Orion. The new combined company will own 40% of the new subsidiary and will also have a first option to exclusive global marketing rights for Orion.

Pixium Vision’s Prima System. Image Credit: Images by Pixium Vision

Image Credit: Boston Scientific
Boston Scientific Corporation received U.S. Food and Drug Administration (FDA) approval of its fourth-generation Vercise Genus™ Deep Brain Stimulation (DBS) System. According to the announcement, The portfolio consists of a family of Bluetooth-enabled, rechargeable and non-rechargeable, MR-conditional, implantable pulse generators (IPGs) that power Cartesia™ Directional Leads.

Impulse Dynamics – the company for which I am CTO and Executive VP – received MR-conditional approval (1.5 T head/limbs with peripheral coils) from both FDA and the European Union.
From the press release:
IMPULSE DYNAMICS ANNOUNCES FDA APPROVAL FOR MAGNETIC RESONANCE IMAGING FDA
Clears Potential Hurdle for Many Heart Failure Patients
MARLTON, N.J.–(BUSINESS WIRE)–Impulse Dynamics, a company dedicated to improving the lives of people with heart failure (HF), today announced the U.S. Food and Drug Administration (FDA) has approved the conditional use of Magnetic Resonance Imaging (MRI) for Optimizer® CCM® delivery systems. This approval represents a significant advance because the population that benefits most from cardiac contractility modulation therapy (patients with moderate to severe HF) often requires advanced diagnostic imaging procedures.

Image Credit: Biotronik
FDA has cleared Biotronik’s Vital Data Sensor to identify body temperature increases potentially associated with fever as part of its new Biomonitor IIIm injectable cardiac monitor (ICM).
The device has shown to achieve a 72 percent reduction in false positives while maintaining 100 percent of AF-related sensitivity. It removes single ectopic beats out of the AF detection algorithm, enhancing diagnostic accuracy.
According to Biotronik, “BIOMONITOR IIIm is the industry leader in battery longevity with 5.5 years of continuous service, providing long-term post-ablation or cryptogenic stroke monitoring. It is also backed by the Patient App, which allows patients to include vital symptom data, providing physicians with symptom-rhythm correlation which strengthens diagnostic precision.”

Image Credit: iota Biosciences
iota Biosciences was established in 2017, building up on the concept of “neural dust” technology invented at the University of California, Berkeley by iota co-founders and co-CEOs Jose Carmena, Ph.D. and Michel Maharbiz, Ph.D.
iota’s “neural dust” consists of a small implantable device (a few mm long) that is powered from an external ultrasound generator. According to iota, their devices can record electrical information, stimulate nerves and communicate with other machines through ultrasound. Iota claims that “neural dust” devices can modulate the information transmitted through nerves, enabling doctors to better treat conditions from arthritis to cardiovascular disease
In 2018 iota completed a $15 million series A funding round aimed at developing a sensing platform. In September 2019, iota entered into a joint R&D agreement with Japanese Astellas Pharma to “design detailed specifications of implantable medical devices and conduct preclinical studies for several diseases with high unmet medical needs.”
Astellas today announced that it would acquire Iota in a deal that includes an initial payment of about $127.5M, covering the equity that Astellas did not acquire in 2018. An additional $176.5M are development-related milestone payments. Iota will be an independent subsidiary under Astellas’ U.S. umbrella.
Eventually, iota hopes to shrink the device to the size of grain of sand that can simultaneously sense neural activity and stimulate nerves to enable highly-targeted closed-loop therapies.
iota Biosciences’ website is at: https://iota.bio/

Impulse Dynamics, Inc., a company dedicated to improving the lives of people with heart failure, today announced its innovative CCM® therapy delivered by the Optimizer® system received an award from the R&D 100 Awards, a prestigious program honoring research and development pioneers and their revolutionary ideas in science and technology.
“Winning this award is a tremendous accomplishment for our company as it represents the recognition by fellow scientists and engineers for the enormous effort that has gone into developing this breakthrough technology. The therapy serves a huge unmet need for a very large group of heart failure patients who suffer every day with their symptoms, so it’s great for the technology to receive recognition at this level,” said David Prutchi, Ph.D., Chief Technology Officer and Executive Vice President at Impulse Dynamics. “I am pleased to congratulate all my colleagues that helped evolve what was initially an intriguing electrophysiological discovery into the transformational therapy that it is today.”
Impulse Dynamics’ website: www.impulse-dynamics.com
Boston Scientific reported today its Q2 2020 results which show a very sharp decline in earnings due to the impact of the Covid-19 pandemic. Specifically for the active implantables industry, CRM dropped from $498M in Q2 2019 to $351M this quarter (-29.4%), and likewise neuromodulation dropped from $204M to $122M (-40%).

Image Credit: CVRx
CVRx’s BeAT-HF phase III randomized clinical trial results were published in the Journal of the American College of Cardiology (“JACC”). Results from the trial were used to obtain FDA PMA for the BAROSTIM NEO device for improvement of symptoms in patients with heart failure.
The main results for the study were:

Image Credit: Abbott
Abbott today announced that the FDA has approved its next-generation Gallant™ line of ICD and CRT-D devices. According to the announcement:
“The devices bring new benefits to patients with heart rhythm disorders, including a patient-preferred design without compromising battery longevity and MRI compatibility. In addition, the new devices offer Bluetooth technology and a new patient smartphone app for improved remote monitoring, allowing for increased patient/physician engagement and streamlined communications.
The new Gallant system pairs with Abbott’s secure myMerlinPulse™, an iOS- and Android™- compatible mobile smartphone app that helps streamline communication between doctors and their patients. The app provides people with access to data, device performance, and transmission history, which helps them take an active role in their healthcare. Through the myMerlinPulse app, physicians can continuously monitor their patients remotely allowing for identification of asymptomatic episodes as well as patient-triggered transmissions, which can lead to earlier intervention and reduce clinical burden.”
Abbott received the CE Mark for these devices back in February 2020.

Image Credit: Boston Scientific
FDA has 510(k)-cleared the LUX-Dx™ Insertable Cardiac Monitor (ICM), which is a long-term diagnostic device implanted in patients to detect arrhythmias associated with conditions such as atrial fibrillation (AF), cryptogenic stroke and syncope
According to the announcement:
“The new LUX-Dx ICM System is designed with a dual-stage algorithm that detects and then verifies potential arrhythmias before an alert is sent to clinicians, thereby providing actionable data for clinical decision-making. Further, the remote programming capabilities of the device via the LATITUDE Clarity™ Data Management System website allow physicians and care teams to adjust event detection settings without requiring an in-person patient appointment, a feature unavailable on any other ICM currently on the market.
…
The dual-stage algorithm within the LUX-Dx ICM System can be programmed to identify AF, atrial flutter, rhythm pause, bradycardia and tachycardia episodes and allows the device to detect arrhythmias each time established thresholds or parameters are exceeded. An additional level of verification filters is then applied, which was developed to catch false positive detections before an alert is sent. Additionally, after implant, patients are provided with a mobile device preloaded with the MyLUX™ app which connects via Bluetooth® to their ICM device. The app transmits device data daily, or as needed, to the LATITUDE Clarity Data Management System giving physicians and care teams timely access to vital information.”
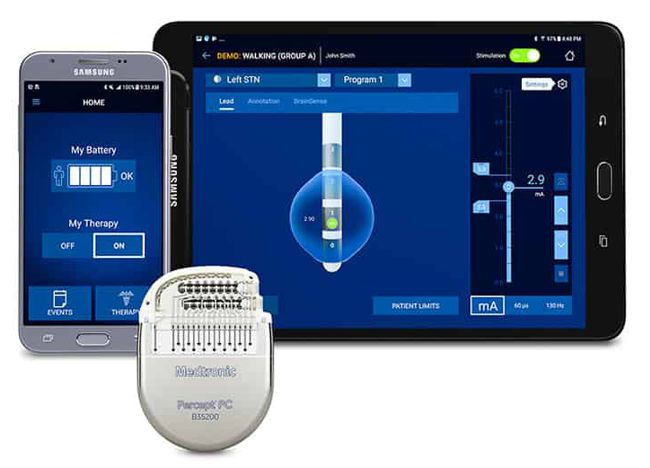
Image Credit: Medtronic
Medtronic announced that it received FDA approval for the Percept™ PC Deep Brain Stimulation (DBS) system.
According to the announcement:
“BrainSense™ technology makes Percept the first and only DBS neurostimulation system with the ability to chronically capture and record brain signals while delivering therapy to patients with neurologic disorders associated with Parkinson’s disease, essential tremor, dystonia, epilepsy or obsessive-compulsive disorder (OCD). Physicians can now track patient brain signals and correlate these with patient-recorded actions or experiences, such as symptoms, side-effects, or medication intake. This enables more personalized, data-driven neurostimulation treatment.
…
In addition to BrainSense technology, the Percept PC DBS system features several leading-edge innovations, including: