
My friend Daniel Villamil from CCC Medical in Uruguay sent me these pictures of a very unique device in his colection. It is a late-1960s/early 1970s pacemaker made in Sao Paulo, Brasil.
UPDATE Oct 3, 2012:
CCC’s CEO Julio Arzuaga recalled that this pacemaker was manufactured in the early 1960s by the Instituto de Cardiologia Dante Pazzanese in Sao Paulo, Brasil. The physicians leading the pacemaker team were Dr. Decio Kormann and Dr. Adib Jatene.
Dr. Orestes Fiandra used to implant these Brasilian pacemakers in Uruguay. However, they were not very reliable. For this reason, and with help from Drs. Kormann and Jatene, Dr. Fiandra started CCC del Uruguay as a more industrial environment for the production of pacemakers.




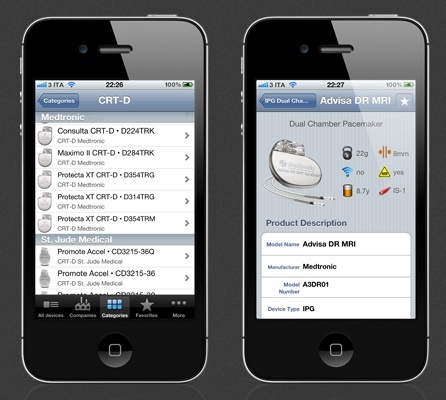
 In 1965, Australian medical device pioneer Noel Gray established Telectronics – Australia’s first manufacturing facility for producing pacemakers that were designed in-house. Telectronics was an innovative developer, achieving some major successes in the early cardiac pacing field, for example, Telectronics’ leads allowed narrowing the pacing pulse to its current nominal of 0.5 milliseconds; encapsulating the pacemaker in titanium instead of epoxy; using a microplasma weld to join the two halves of the pacemaker capsule; creating one of the first rate-responsive ‘demand’ pacemakers; and isolating the pacemaker’s battery in a separate compartment to deal with the problem of leaking mercury-zinc batteries.
In 1965, Australian medical device pioneer Noel Gray established Telectronics – Australia’s first manufacturing facility for producing pacemakers that were designed in-house. Telectronics was an innovative developer, achieving some major successes in the early cardiac pacing field, for example, Telectronics’ leads allowed narrowing the pacing pulse to its current nominal of 0.5 milliseconds; encapsulating the pacemaker in titanium instead of epoxy; using a microplasma weld to join the two halves of the pacemaker capsule; creating one of the first rate-responsive ‘demand’ pacemakers; and isolating the pacemaker’s battery in a separate compartment to deal with the problem of leaking mercury-zinc batteries. 



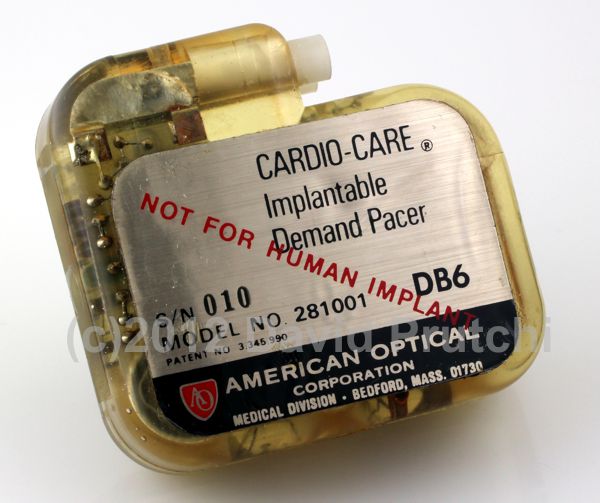




 One of the indicators of metabolic demand that has been used for controlling the rate of pacemakers is central venous blood temperature (CVT).
One of the indicators of metabolic demand that has been used for controlling the rate of pacemakers is central venous blood temperature (CVT).