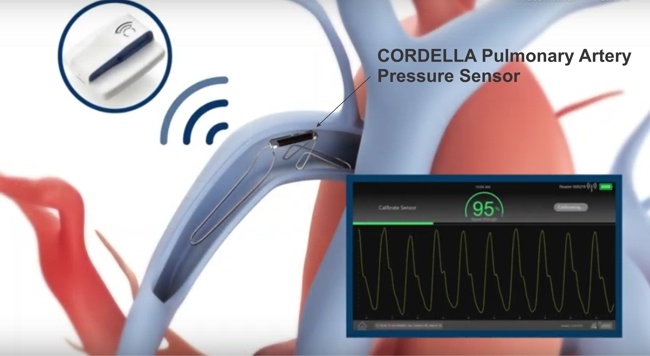
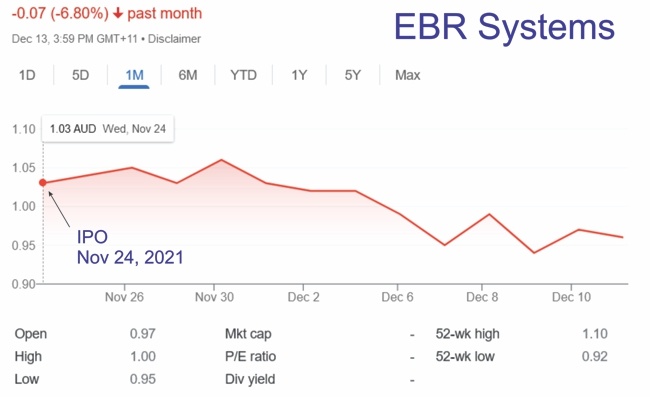
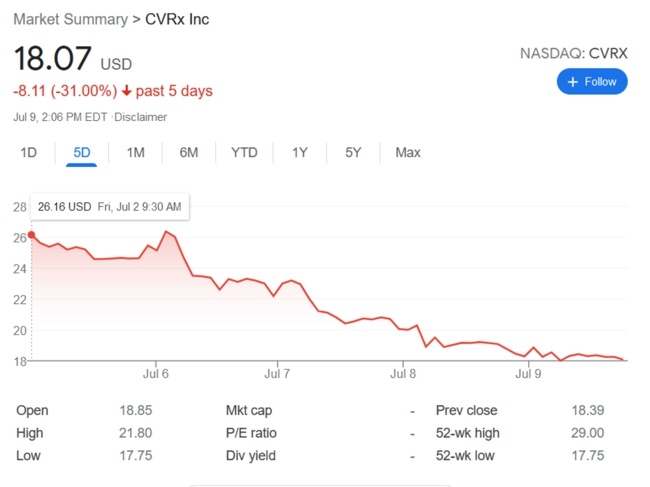
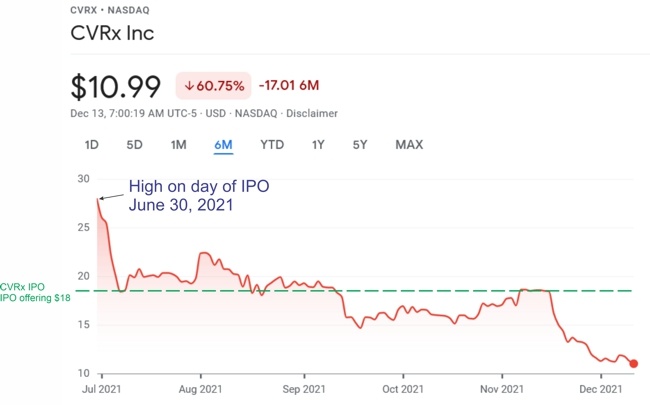
I’ll be presenting the keynote “AIMD-based Therapies of Heart Failure” at the First Workshop on Active Implantable Medical Devices dedicated to understanding the trends and challenges in bioelectronic medicine.
For the complete announcement of the workshop with some great talks by Rikky Muller (UC Berkeley), Pedro Arzuaga (Impulse Dynamics), Marcelo Barú (Biotronik), Federico De Mula (Integer), and Victor Pikov (Medipace) click here: AIMD Workshop LASCAS 2024 Program.