 Yesterday Boston Scientific announced financial results for the first quarter ended March 31, 2012. Sales of Cardiac Rhythm Management devices were $501M vs. $559M for Q1 a year ago, or a decrease of 10%. Sales of Neuromodulation devices increased by 8% a year ago from $77M to $84M for Q1.
Yesterday Boston Scientific announced financial results for the first quarter ended March 31, 2012. Sales of Cardiac Rhythm Management devices were $501M vs. $559M for Q1 a year ago, or a decrease of 10%. Sales of Neuromodulation devices increased by 8% a year ago from $77M to $84M for Q1.
St. Jude’s Reports Q1 2012 AIMD Sales Results: CRM down 4%, Neuromodulation up 12%
 St. Jude Medical today reported sales and net earnings for the first quarter ended March 31, 2012.
St. Jude Medical today reported sales and net earnings for the first quarter ended March 31, 2012.
Total CRM sales, which include ICD and pacemaker products, were $735 million for the first quarter of 2012, a 4 percent decrease compared with the first quarter of 2011. Of that total, ICD product sales were $450 million in the first quarter, a 3 percent decrease compared with the first quarter of 2011. First quarter pacemaker sales were $285 million, a decrease of 4 percent from the comparable quarter of 2011.
St. Jude Medical sales of neuromodulation products were $103 million in the first quarter of 2012, up 12 percent from the comparable quarter of 2011.
St. Jude Starts Trial of Accent MRI Pacemaker and Tendril MRI Lead

Image Credit: St. Jude Medical
Today St. Jude announced the first implant in its Accent MRI(R) Pacemaker and Tendril MRI(R) Lead IDE Study (MRI Study). The ultimate goal of the study is to determine if patients with these devices can safely undergo full-body, high resolution Magnetic Resonance Imaging (MRI) scans to better accommodate their medical needs. The investigational Accent MRI Pacemaker system from St. Jude Medical offers an advanced pacing platform that provides wireless telemetry and algorithms to help address individual patient conditions.
Digikon’s History
 In response to my post “A Challenge to History Buffs: Who Was Digikon?“, Paolo Pagani sent me the following message:
In response to my post “A Challenge to History Buffs: Who Was Digikon?“, Paolo Pagani sent me the following message:
“Digikon was in the years 1977-1985 the brand name product in Italy by Biotec Biomedical Technologies of Bologna – ITALY.
Pacemakers were a Digikon O.E.M. production for the trading company of Milan Italy KONTRON already a distributor in Italy of Medtronic.
Biotec developed the first pacemaker VVIR based on physiological changes in thoracic impedance due to respiration. (Biotec RDP-3)
Biotec-Bologna was acquired by Medtronic in August 1985.”
Thank you Paolo!
Medtronic Receives FDA Appoval for Expanded Use of CRT-D to Class II HF
 Medtronic received FDA approval for the expanded use of CRT-D in mildly-symptomatic heart failure patients. The expanded indication includes New York Heart Association (NYHA) Class II heart failure patients with a left ventricular ejection fraction (LVEF) of less than or equal to 30 percent, left bundle branch block (LBBB), and a QRS duration greater than or equal to 130 milliseconds. Nearly 200,000 Americans are considered NYHA Class II, with another 620,000 people worldwide fitting this designation.
Medtronic received FDA approval for the expanded use of CRT-D in mildly-symptomatic heart failure patients. The expanded indication includes New York Heart Association (NYHA) Class II heart failure patients with a left ventricular ejection fraction (LVEF) of less than or equal to 30 percent, left bundle branch block (LBBB), and a QRS duration greater than or equal to 130 milliseconds. Nearly 200,000 Americans are considered NYHA Class II, with another 620,000 people worldwide fitting this designation.
The FDA’s decision for the expanded indication rests on data from the pivotal REVERSE (REsynchronization reVErses Remodeling in Systolic left vEntricular dysfunction) and landmark RAFT (Resynchronization/Defibrillation in Ambulatory Heart Failure Trial) clinical trials, which showed that CRT-D can benefit mildly symptomatic heart failure patients by reducing mortality and heart failure hospitalization rates.The expanded indication includes New York Heart Association (NYHA) Class II heart failure patients with a left ventricular ejection fraction (LVEF) of less than or equal to 30 percent, left bundle branch block (LBBB), and a QRS duration greater than or equal to 130 milliseconds. Nearly 200,000 Americans are considered NYHA Class II, with another 620,000 people worldwide fitting this designation.
Click here for press release from Medtronic.
Monash University in Australia Starts Test of Direct-to-Brain Visual Prosthesis Chips

Image Credit: Monash Vision Group, Monash University, Australia
Engineers from the Monash Vision Group (MVG) have begun trialling the ASICs for a direct-to-brain visual prosthesis that is expected to enter human clinical trials in 2014.
The prosthesis will consist of a tiny camera mounted into a pair of glasses, which acts as the retina; a pocket processor, which takes the electronic information from the camera and converts it into signals enabling the brain to build up a visual construct; and cortical implants of several tiles which will be the portal for the stimulation of the visual cortex. Continue reading
Magnetically-Coupled Motor/Generator Transfers 100 mW to Implant at 50 cm Range
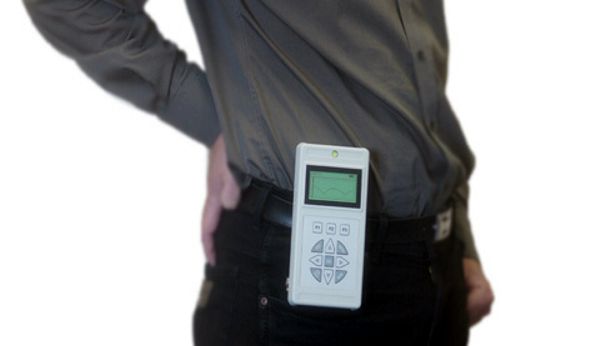
Image Credit: Fraunhofer Institute for Ceramic Technologies and Systems
Scientists at the Fraunhofer Institute for Ceramic Technologies and Systems developed a magnetically-coupled motor/generator system that they claim is able to transcutaneously transfer 100 mW to an implant up to 50 cm away.
In the external power-transfer module, a rotating magnet driven by an EC motor generates a magnetic rotary field. A magnetic pellet in the implanted receiver connects to the alternating exterior magnetic field and as a result, is set in rotation itself. The rotational movement is transformed into electricity, thus the power is produced right in the generator module. “With magnetic coupling, power can be transported through all non-magnetic materials, such as biological tissue, bones, organs, water, plastic or even a variety of metals. Moreover, the magnetic field produced has no harmful side effects on humans. It doesn‘t even heat up tissue,” says Dr. Holger Lausch, highlighting the advantages of the system. Continue reading
John Hopkins Researcher Develops New Early-Warning Seizures Detector with Low False-Positive Rate
 Johns Hopkins’ Sridevi V. Sarma, an assistant professor of biomedical engineering, has devised new seizure detection software that, in early testing, significantly cuts the number of unneeded brain-stimulation therapy that an epilepsy patient would receive.
Johns Hopkins’ Sridevi V. Sarma, an assistant professor of biomedical engineering, has devised new seizure detection software that, in early testing, significantly cuts the number of unneeded brain-stimulation therapy that an epilepsy patient would receive.
According to Sarma, “These devices use algorithms—a series of mathematical steps—to figure out when to administer the treatment,” Sarma said. “They’re very good at detecting when a seizure is about to happen, but they also produce lots of false positives, sometimes hundreds in one day. If you introduce electric current to the brain too often, we don’t know what the health impacts might be. Also, too many false alarms can shorten the life of the battery that powers the device, which must be replaced surgically.” Continue reading
St. Jude Seeks Retraction of Heart Rhythm Journal Manuscript by Dr. Robert Hauser Regarding the Failure of Riata and Riata ST Implantable Cardioverter-Defibrillator Leads
 St. Jude announced it is seeking a retraction of the manuscript accepted for publication in the Heart Rhythm Journal by Dr. Robert Hauser, et al., titled, “Deaths Caused by the Failure of Riata and Riata ST Implantable Cardioverter-Defibrillator Leads.” St. Jude claims that the research performed by Hauser undercounted and excluded MAUDE data reports for Medtronic product resulting in substantial factual errors.
St. Jude announced it is seeking a retraction of the manuscript accepted for publication in the Heart Rhythm Journal by Dr. Robert Hauser, et al., titled, “Deaths Caused by the Failure of Riata and Riata ST Implantable Cardioverter-Defibrillator Leads.” St. Jude claims that the research performed by Hauser undercounted and excluded MAUDE data reports for Medtronic product resulting in substantial factual errors.
According to the press release: Continue reading
St. Jude Warns Physicians of Potential Problem with QuickSite and QuickFlex LV CRT Leads

Image Credit: St. Jude Medical
St. Jude announced it is proactively informing physicians about visual observations of externalized conductors on the silicone end of QuickSite® and QuickFlex® Left-Ventricular Leads, used to connect Cardiac Resynchronization Therapy devices to the heart.
The announcement noted that there have been no reports of patient injury or loss of therapy due to externalized conductors in these leads, but as a conservative measure, St. Jude Medical is communicating with physicians about the incidence rate so they have the most updated lead performance information with which to make important patient care decisions.
According to the announcement, “St. Jude Medical has confirmed 39 cases of externalized conductors, out of 171,000 QuickSite and QuickFlex leads sold worldwide, resulting in a current reported incidence rate of 0.023 percent, or 2.3 in 10,000. Because these leads continue to function normally, the company expects that this rate is under-reported. Based on an analysis of leads returned to the company and recent fluoroscopic images of implanted leads still in clinical use, St. Jude Medical estimates that 3 to 4 percent of QuickSite and QuickFlex leads may exhibit externalized conductors. As a result of this estimated rate, the company felt it was prudent to communicate with physicians about the externalized conductors at this time.”
St. Jude also announced they will no longer sell these lead models.
St. Jude Medical Announces Clinical Benefits of Quadripolar CRT Pacing at ACC

Image Credit: St. Jude Medical
St. Jude Medical announced that clinical findings on quadripolar pacing will be presented at the 61st Annual Scientific Sessions of the American College of Cardiology (ACC, Chicago, March 24-27 2012).
According to St. Jude’s announcement, quadripolar pacing allows physicians the ability to use multisite left-ventricular (LV) pacing. Studies that examined the role of multisite pacing in improving hemodynamics and reducing dyssynchrony as compared to traditional bi-ventricular pacing will be presented in the following two posters:
FDA AIMD PMA/Supplement Approvals for January
Cool Reed Switch Manufacturing Video

Magnetic reed switches are used in active implantable medical devices as a simple way of placing the device in a known operational mode when a programmer is not available. For example, placing a magnet on a pacemaker sets it to VVI mode with a manufacturer-specified set of parameters. In other devices (e.g. implantable cardioverter/defibrillators), placing a magnet on the IPG inhibits the delivery of high voltage defibrillation.
Although the reed switches that I use don’t come from Meder Electronics, I came across a very cool video that Meder produced regarding their manufacturing process. Continue reading
ImThera Receives CE Mark for the Aura6000 System to Treat Obstructive Sleep Apnea
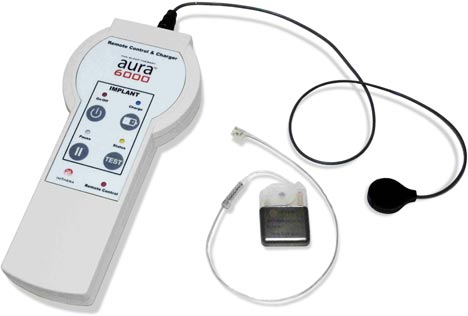
Image Credit: Imthera
ImThera Medical announced this week that it received the CE Mark for the Aura6000 System to Treat Obstructive Sleep Apnea. Continue reading
Boston Scientific to Acquire Cameron Health for $150M Up-Front + $150M Upon FDA Approval

Image Credit: Boston Scientific
Boston Scientific announced the exercise of its option to acquire Cameron Health. Cameron Health developed the world’s first and only commercially-available subcutaneous implantable cardioverter defibrillator – the S-ICD® System that I blogged about a few weeks ago.
The agreement calls for an upfront payment of $150 million, payable upon transaction closing, an additional potential $150 million payment upon FDA approval of the S-ICD System, plus up to an additional $1.050 billion of potential payments upon achievement of specified revenue-based milestones over a six-year period following FDA approval. Closing of the transaction is subject to customary conditions, including relevant antitrust clearance, and is expected to occur in the second or third quarter of 2012.

