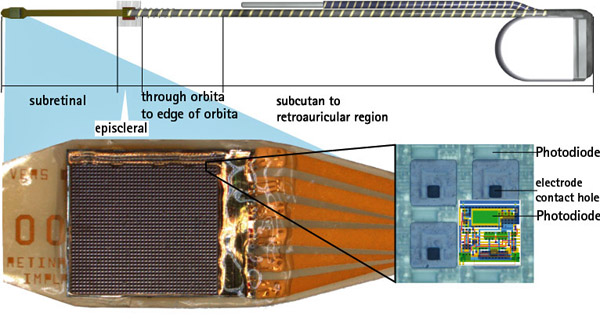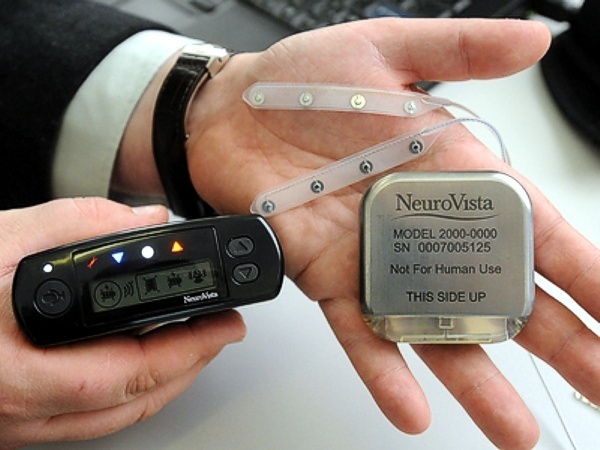
Seattle-based NeuroVista was founded in 2002 by Dr. Daniel DiLorenzo to develop an implantable device for the early detection of epileptic seizures. The NeuroVista seizure advisory system is based on an implantable device that senses EEG irregularities that precede a seizure. Early warning allows patients to take medicine and find a safe place to lie down. Although some epilepsy sufferers can feel seizures coming, many cannot.
In NeuroVista’s Seizure Advisory System (SAS), intracranial EEG signals are recorded through electrodes implanted between the skull and the brain surface. Data storage and signal telemetry takes place within the pectorally-implanted can that transmits signals wirelessly to an external handheld device that processes the data and transmits visual and audible signals to the patient. The external pager-like receiver displays a blue light when there is a low likelihood of seizures, white indicates medium susceptibility, and red alerts to a high likelihood of impending seizure.
Results of the system on 11 patients were published in the Lancet an a paper entitled “Prediction of seizure likelihood with a long-term, implanted seizure advisory system in patients with drug-resistant epilepsy: a first-in-man study.” From the paper:

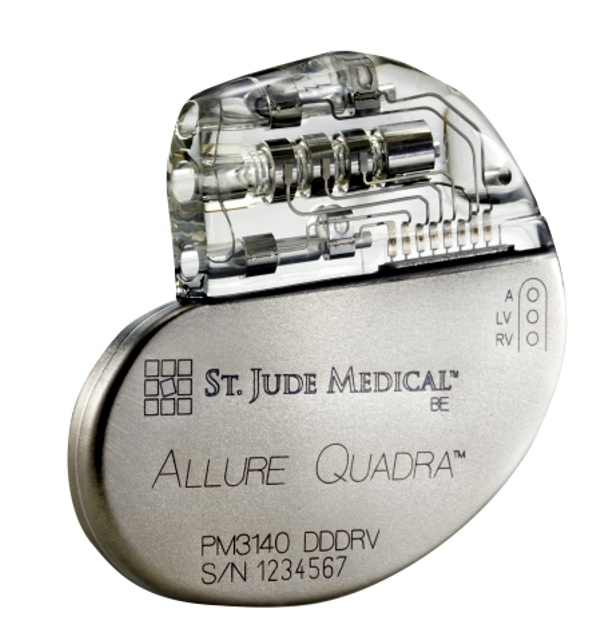 St. Jude Medical today announced CE Mark approval and European launch of its Allure Quadra™ Cardiac Resynchronization Therapy Pacemaker (CRT-P), which brings the quadripolar lead technology to the pacemaker market for the first time. According to the press release:
St. Jude Medical today announced CE Mark approval and European launch of its Allure Quadra™ Cardiac Resynchronization Therapy Pacemaker (CRT-P), which brings the quadripolar lead technology to the pacemaker market for the first time. According to the press release:


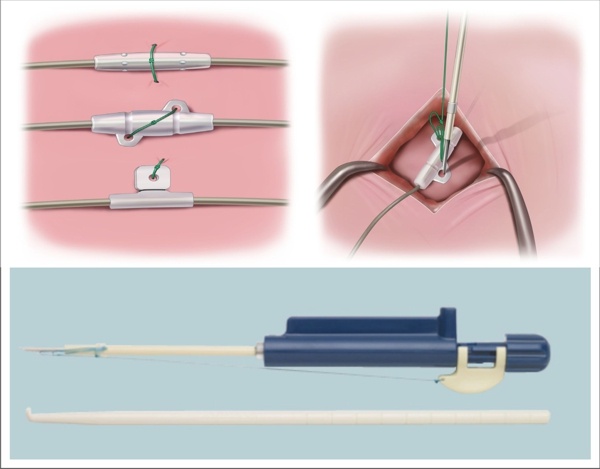
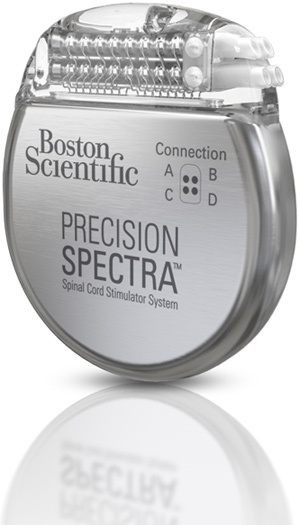



 Biotronik announced that it had received CE-approval for the world’s first DF4 ICD/CRT-D series approved for MRI. In addition, this series contains one of the world’s smallest ICDs– the Iforia single chamber ICD.
Biotronik announced that it had received CE-approval for the world’s first DF4 ICD/CRT-D series approved for MRI. In addition, this series contains one of the world’s smallest ICDs– the Iforia single chamber ICD.