
Swiss medical device manufacturer Sequana Medical developed the ALFApump® System to pump fluids accummulated in the abdomen of a patient into the bladder.

Swiss medical device manufacturer Sequana Medical developed the ALFApump® System to pump fluids accummulated in the abdomen of a patient into the bladder.

Image Source: Nevro's Website
Nevro Corp announced that FDA has granted approval for initiation of its SENZA-RCT study, a U.S. prospective, randomized, controlled pivotal clinical trial evaluating the safety and efficacy of Nevro’s high-frequency spinal cord stimulation system for the treatment of chronic pain.

Image Credit: St. Jude Medical
St. Jude Medical today announced FDA approval of its Assura™ portfolio of implantable cardioverter defibrillators (ICDs) and cardiac resynchronization therapy defibrillators (CRT-Ds).The new implantable defibrillators feature SecureSense™ RV Lead Noise Discrimination, an algorithm that expands the St. Jude Medical ShockGuard® Technology and offers advanced sensing options designed to reduce the incidence of inappropriate shocks for patients with these devices.
According to St. Jude’s announcement:
 Today EnteroMedics recorded revenue for the first time since it was incorporated nearly eight years ago. The company reported revenue of about $123,000 in the first quarter of the year from the sale of its Maestro RC implantable vagus nerve stimulation system for treating obesity. Revenue was generated through sales by its distribution partner in Australia.
Today EnteroMedics recorded revenue for the first time since it was incorporated nearly eight years ago. The company reported revenue of about $123,000 in the first quarter of the year from the sale of its Maestro RC implantable vagus nerve stimulation system for treating obesity. Revenue was generated through sales by its distribution partner in Australia.
This is an important step for an implantable device company that faced very tough times in 2009 after its US clinical trial failed to meet a critical effectiveness goal. EnteroMedics is currently conducting a pivotal trial that is expected to end in Q4 2012. EnteroMedics expects to file a premarket approval application with the FDA in the first half of 2013 if the data from the trial is positive.
 Yesterday Boston Scientific announced financial results for the first quarter ended March 31, 2012. Sales of Cardiac Rhythm Management devices were $501M vs. $559M for Q1 a year ago, or a decrease of 10%. Sales of Neuromodulation devices increased by 8% a year ago from $77M to $84M for Q1.
Yesterday Boston Scientific announced financial results for the first quarter ended March 31, 2012. Sales of Cardiac Rhythm Management devices were $501M vs. $559M for Q1 a year ago, or a decrease of 10%. Sales of Neuromodulation devices increased by 8% a year ago from $77M to $84M for Q1.
 St. Jude Medical today reported sales and net earnings for the first quarter ended March 31, 2012.
St. Jude Medical today reported sales and net earnings for the first quarter ended March 31, 2012.
Total CRM sales, which include ICD and pacemaker products, were $735 million for the first quarter of 2012, a 4 percent decrease compared with the first quarter of 2011. Of that total, ICD product sales were $450 million in the first quarter, a 3 percent decrease compared with the first quarter of 2011. First quarter pacemaker sales were $285 million, a decrease of 4 percent from the comparable quarter of 2011.
St. Jude Medical sales of neuromodulation products were $103 million in the first quarter of 2012, up 12 percent from the comparable quarter of 2011.

Image Credit: St. Jude Medical
Today St. Jude announced the first implant in its Accent MRI(R) Pacemaker and Tendril MRI(R) Lead IDE Study (MRI Study). The ultimate goal of the study is to determine if patients with these devices can safely undergo full-body, high resolution Magnetic Resonance Imaging (MRI) scans to better accommodate their medical needs. The investigational Accent MRI Pacemaker system from St. Jude Medical offers an advanced pacing platform that provides wireless telemetry and algorithms to help address individual patient conditions.
 In response to my post “A Challenge to History Buffs: Who Was Digikon?“, Paolo Pagani sent me the following message:
In response to my post “A Challenge to History Buffs: Who Was Digikon?“, Paolo Pagani sent me the following message:
“Digikon was in the years 1977-1985 the brand name product in Italy by Biotec Biomedical Technologies of Bologna – ITALY.
Pacemakers were a Digikon O.E.M. production for the trading company of Milan Italy KONTRON already a distributor in Italy of Medtronic.
Biotec developed the first pacemaker VVIR based on physiological changes in thoracic impedance due to respiration. (Biotec RDP-3)
Biotec-Bologna was acquired by Medtronic in August 1985.”
Thank you Paolo!
 St. Jude announced it is seeking a retraction of the manuscript accepted for publication in the Heart Rhythm Journal by Dr. Robert Hauser, et al., titled, “Deaths Caused by the Failure of Riata and Riata ST Implantable Cardioverter-Defibrillator Leads.” St. Jude claims that the research performed by Hauser undercounted and excluded MAUDE data reports for Medtronic product resulting in substantial factual errors.
St. Jude announced it is seeking a retraction of the manuscript accepted for publication in the Heart Rhythm Journal by Dr. Robert Hauser, et al., titled, “Deaths Caused by the Failure of Riata and Riata ST Implantable Cardioverter-Defibrillator Leads.” St. Jude claims that the research performed by Hauser undercounted and excluded MAUDE data reports for Medtronic product resulting in substantial factual errors.
According to the press release: Continue reading→

Image Credit: St. Jude Medical
St. Jude announced it is proactively informing physicians about visual observations of externalized conductors on the silicone end of QuickSite® and QuickFlex® Left-Ventricular Leads, used to connect Cardiac Resynchronization Therapy devices to the heart.
The announcement noted that there have been no reports of patient injury or loss of therapy due to externalized conductors in these leads, but as a conservative measure, St. Jude Medical is communicating with physicians about the incidence rate so they have the most updated lead performance information with which to make important patient care decisions.
According to the announcement, “St. Jude Medical has confirmed 39 cases of externalized conductors, out of 171,000 QuickSite and QuickFlex leads sold worldwide, resulting in a current reported incidence rate of 0.023 percent, or 2.3 in 10,000. Because these leads continue to function normally, the company expects that this rate is under-reported. Based on an analysis of leads returned to the company and recent fluoroscopic images of implanted leads still in clinical use, St. Jude Medical estimates that 3 to 4 percent of QuickSite and QuickFlex leads may exhibit externalized conductors. As a result of this estimated rate, the company felt it was prudent to communicate with physicians about the externalized conductors at this time.”
St. Jude also announced they will no longer sell these lead models.

Image Credit: St. Jude Medical
St. Jude Medical announced that clinical findings on quadripolar pacing will be presented at the 61st Annual Scientific Sessions of the American College of Cardiology (ACC, Chicago, March 24-27 2012).
According to St. Jude’s announcement, quadripolar pacing allows physicians the ability to use multisite left-ventricular (LV) pacing. Studies that examined the role of multisite pacing in improving hemodynamics and reducing dyssynchrony as compared to traditional bi-ventricular pacing will be presented in the following two posters:
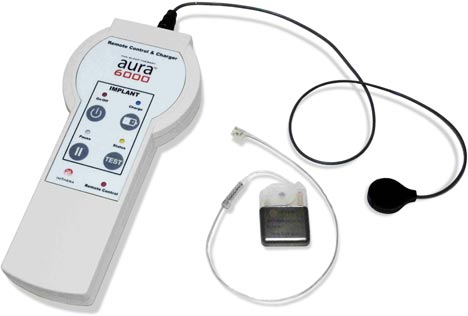
Image Credit: Imthera
ImThera Medical announced this week that it received the CE Mark for the Aura6000 System to Treat Obstructive Sleep Apnea. Continue reading→

Image Credit: Boston Scientific
Boston Scientific announced the exercise of its option to acquire Cameron Health. Cameron Health developed the world’s first and only commercially-available subcutaneous implantable cardioverter defibrillator – the S-ICD® System that I blogged about a few weeks ago.
The agreement calls for an upfront payment of $150 million, payable upon transaction closing, an additional potential $150 million payment upon FDA approval of the S-ICD System, plus up to an additional $1.050 billion of potential payments upon achievement of specified revenue-based milestones over a six-year period following FDA approval. Closing of the transaction is subject to customary conditions, including relevant antitrust clearance, and is expected to occur in the second or third quarter of 2012.
Image Credit: Medtronic
Medtronic today announced the receipt of CE Mark and launch of the CapSure Sense MRI™ SureScan® pacing leads, which are approved for use during MRI procedures. The newly approved leads are the smallest MR–Conditional leads available in the world with a 5.3 French isodiametric lead body. The new leads are passive-fixation leads. Previously approved Medtronic MR–Conditional leads are active fixation leads.
Click here for Medtronic’s product page for the CapSure Sense MRI™ SureScan® Pacing Leads.
 In 1965, Australian medical device pioneer Noel Gray established Telectronics – Australia’s first manufacturing facility for producing pacemakers that were designed in-house. Telectronics was an innovative developer, achieving some major successes in the early cardiac pacing field, for example, Telectronics’ leads allowed narrowing the pacing pulse to its current nominal of 0.5 milliseconds; encapsulating the pacemaker in titanium instead of epoxy; using a microplasma weld to join the two halves of the pacemaker capsule; creating one of the first rate-responsive ‘demand’ pacemakers; and isolating the pacemaker’s battery in a separate compartment to deal with the problem of leaking mercury-zinc batteries. Continue reading→
In 1965, Australian medical device pioneer Noel Gray established Telectronics – Australia’s first manufacturing facility for producing pacemakers that were designed in-house. Telectronics was an innovative developer, achieving some major successes in the early cardiac pacing field, for example, Telectronics’ leads allowed narrowing the pacing pulse to its current nominal of 0.5 milliseconds; encapsulating the pacemaker in titanium instead of epoxy; using a microplasma weld to join the two halves of the pacemaker capsule; creating one of the first rate-responsive ‘demand’ pacemakers; and isolating the pacemaker’s battery in a separate compartment to deal with the problem of leaking mercury-zinc batteries. Continue reading→