 WISE (Wiringless Implantable Stretchable Electronics), a Milan- and Berlin-based medical device companyraised a seed-round of € 1 M to advance the development of its first product aimed at the spinal cord stimulation market.
WISE (Wiringless Implantable Stretchable Electronics), a Milan- and Berlin-based medical device companyraised a seed-round of € 1 M to advance the development of its first product aimed at the spinal cord stimulation market.
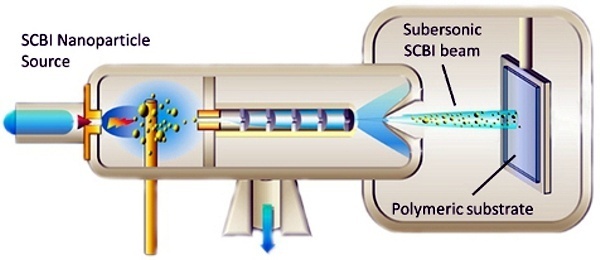
WISE’s technology relies on Supersonic Cluster Beam Implantation (SCBI) for the metallization of polymers and elastomers. SCBI allows to fabricate complex microelectronic circuits and interconnects on stretchable and conformable supports that preserve their electrical performances after extensive cycles of stretching and bending of the substrates. The devices can be efficiently fabricated on biocompatible platforms, and are therefore suitable for the production of implantable biomedical devices.



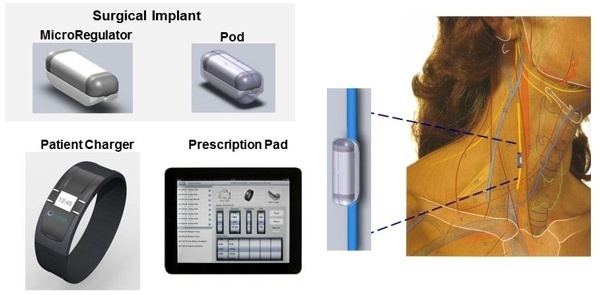
 Neuros Medical received an Investigational Device Exemption to conduct a pivotal clinical trial to evaluate the Altius™ System High Frequency Nerve Block technology for the management of intractable limb pain of amputees. The prospective, randomized, controlled pivotal clinical trial will consist of 130 patients at 15 institutions in the U.S. to evaluate the safety and efficacy of Neuros Medical’s Altius System.
Neuros Medical received an Investigational Device Exemption to conduct a pivotal clinical trial to evaluate the Altius™ System High Frequency Nerve Block technology for the management of intractable limb pain of amputees. The prospective, randomized, controlled pivotal clinical trial will consist of 130 patients at 15 institutions in the U.S. to evaluate the safety and efficacy of Neuros Medical’s Altius System.
 Net product sales increased 12.4% to $67.4 million in the first fiscal quarter ended July 26 for Cyberonics, Inc. of Houston, TX. Including license revenue, sales were up 14.2% overall, with Europe in particular contributing a strong performance. Diluted earnings per share were adjusted by $0.17 cents due to a litigation settlement.
Net product sales increased 12.4% to $67.4 million in the first fiscal quarter ended July 26 for Cyberonics, Inc. of Houston, TX. Including license revenue, sales were up 14.2% overall, with Europe in particular contributing a strong performance. Diluted earnings per share were adjusted by $0.17 cents due to a litigation settlement.
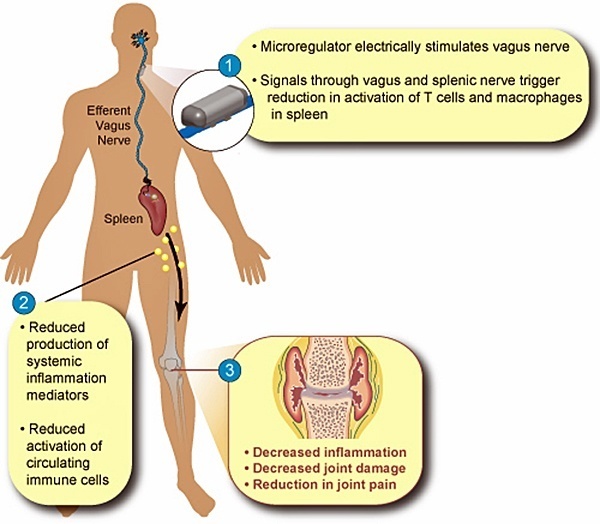

 Start-up company
Start-up company  Boston Scientific announced that it has launched a clinical trial to determine whether occipital nerve stimulation (ONS) using the Precision™ System can safely and effectively treat chronic migraine when used in conjunction with anti-migraine medications. According to the press release:
Boston Scientific announced that it has launched a clinical trial to determine whether occipital nerve stimulation (ONS) using the Precision™ System can safely and effectively treat chronic migraine when used in conjunction with anti-migraine medications. According to the press release: Greatbatch Medical, which moved its headquarters to the Dallas, TX area last year, announced that it has set a target of at least 5%/yr organic growth. To accomplish this growth, the Company recently
Greatbatch Medical, which moved its headquarters to the Dallas, TX area last year, announced that it has set a target of at least 5%/yr organic growth. To accomplish this growth, the Company recently