St. Jude Medical today announced the completion of its acquisition of Nanostim, Inc., a privately-owned developer of miniaturized, leadless pacemakers. The acquisition adds the world’s first and only leadless pacemaker to the St. Jude Medical product portfolio.
According to the press release:
Unlike conventional pacemakers that require a more invasive surgery, the Nanostim™ leadless pacemaker is designed to be implanted directly into the heart via a minimally invasive procedure. The device is delivered using a steerable catheter through the femoral vein, eliminating the need to surgically create a pocket for the pacemaker and insulated wires (called leads) that have historically been recognized as the most vulnerable component of pacing systems. The Nanostim leadless pacemaker recently received CE Mark approval and will be available soon in select European markets. It also recently received U.S. Food and Drug Administration (FDA) conditional approval for its Investigational Device Exemption (IDE) application and pivotal clinical trial protocol to begin evaluating Nanostim leadless technology in the U.S.





 Net product sales increased 12.4% to $67.4 million in the first fiscal quarter ended July 26 for Cyberonics, Inc. of Houston, TX. Including license revenue, sales were up 14.2% overall, with Europe in particular contributing a strong performance. Diluted earnings per share were adjusted by $0.17 cents due to a litigation settlement.
Net product sales increased 12.4% to $67.4 million in the first fiscal quarter ended July 26 for Cyberonics, Inc. of Houston, TX. Including license revenue, sales were up 14.2% overall, with Europe in particular contributing a strong performance. Diluted earnings per share were adjusted by $0.17 cents due to a litigation settlement.

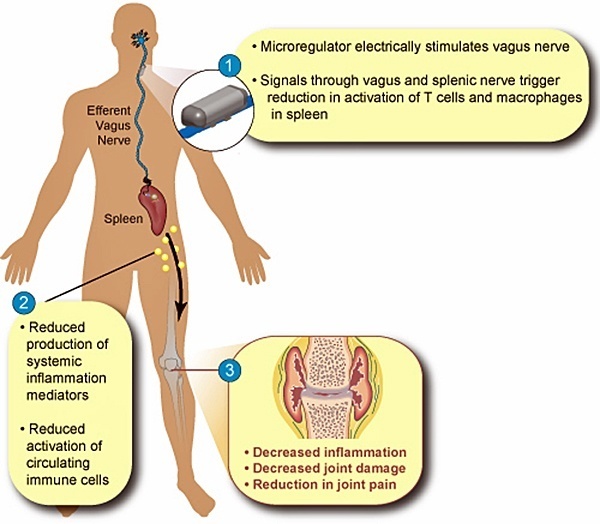


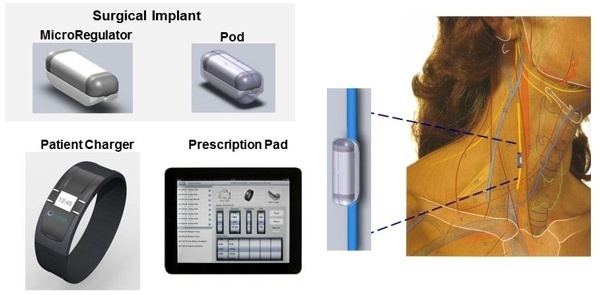
 Start-up company
Start-up company