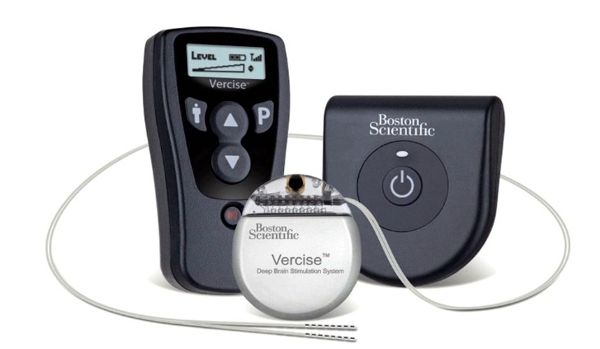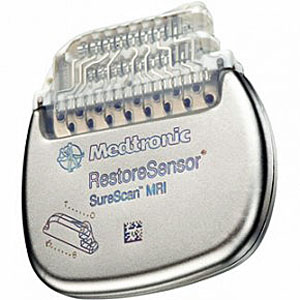
Medtronic has introduced in Europe the first and only implantable neurostimulation systems indicated for use in the treatment of chronic back and/or leg pain that are designed for full-body Magnetic Resonance Imaging (MRI) scans under specific conditions. Medtronic SureScan neurostimulation systems include enhancements to existing devices as well as specially designed leads to reduce or eliminate the hazards produced by the MRI environment. The devices also include a proprietary SureScan programming feature, which sets the device into an appropriate mode for the MRI environment.


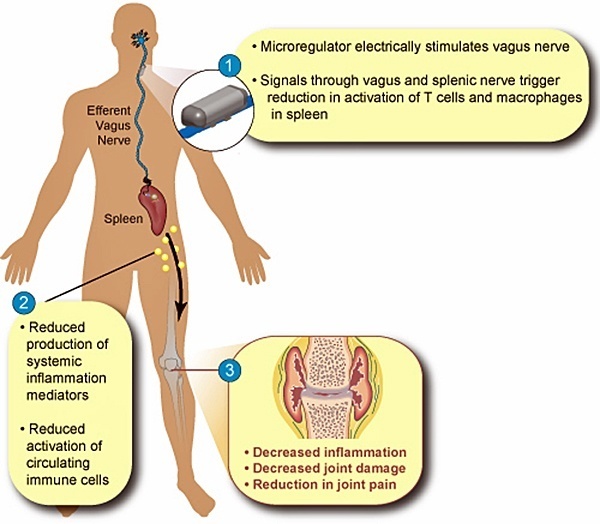
 Toronto-based
Toronto-based 

 I received today a
I received today a 
 Biotronik announced the European market release of BioMonitor®, an implantable cardiac device designed for the highly accurate and reliable monitoring and management of patients with atrial fibrillation (AF) or unexplained syncope.
Biotronik announced the European market release of BioMonitor®, an implantable cardiac device designed for the highly accurate and reliable monitoring and management of patients with atrial fibrillation (AF) or unexplained syncope.