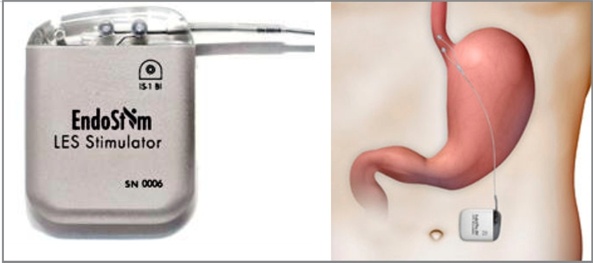
EndoStim announced that Argentina’s National Administration of Drugs, Food and Medical Technology (ANMAT) approved its Lower Esophageal Sphincter (LES) Stimulation System for Gastroesophageal Reflux Disease (GERD). EndoStim is currently available in Europe, Asia and several other countries in Latin America such as Chile and Colombia.










 Neuros Medical received an Investigational Device Exemption to conduct a pivotal clinical trial to evaluate the Altius™ System High Frequency Nerve Block technology for the management of intractable limb pain of amputees. The prospective, randomized, controlled pivotal clinical trial will consist of 130 patients at 15 institutions in the U.S. to evaluate the safety and efficacy of Neuros Medical’s Altius System.
Neuros Medical received an Investigational Device Exemption to conduct a pivotal clinical trial to evaluate the Altius™ System High Frequency Nerve Block technology for the management of intractable limb pain of amputees. The prospective, randomized, controlled pivotal clinical trial will consist of 130 patients at 15 institutions in the U.S. to evaluate the safety and efficacy of Neuros Medical’s Altius System.


