
On January 20, 2014, Mass Device reported that Greatbatch filed a CE Mark application for its spinal cord stimulation system with German notified body TÜV SÜD America. The spinal cord stimulation system comes from Greatbatch’s subsidiary, QiG Group. The company also applied for pre-market approval last month.


![IMAGE_Nanostim_Product_Shot[1]](https://www.implantable-device.com/wp-content/uploads/2013/10/IMAGE_Nanostim_Product_Shot1.jpg)







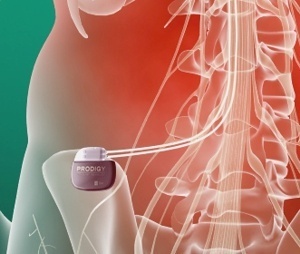 St. Jude announced today that it has initiated a clinical study of the Prodigy™ neurostimulator, which is the first SCS system able to deliver a proprietary mode of stimulation therapy called burst stimulation.
St. Jude announced today that it has initiated a clinical study of the Prodigy™ neurostimulator, which is the first SCS system able to deliver a proprietary mode of stimulation therapy called burst stimulation.
 A
A