Image Credit: Medtronic
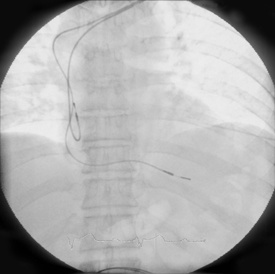
Medtronic today announced the receipt of CE Mark and launch of the CapSure Sense MRI™ SureScan® pacing leads, which are approved for use during MRI procedures. The newly approved leads are the smallest MR–Conditional leads available in the world with a 5.3 French isodiametric lead body. The new leads are passive-fixation leads. Previously approved Medtronic MR–Conditional leads are active fixation leads.
Click here for Medtronic’s product page for the CapSure Sense MRI™ SureScan® Pacing Leads.

 In 1965, Australian medical device pioneer Noel Gray established Telectronics – Australia’s first manufacturing facility for producing pacemakers that were designed in-house. Telectronics was an innovative developer, achieving some major successes in the early cardiac pacing field, for example, Telectronics’ leads allowed narrowing the pacing pulse to its current nominal of 0.5 milliseconds; encapsulating the pacemaker in titanium instead of epoxy; using a microplasma weld to join the two halves of the pacemaker capsule; creating one of the first rate-responsive ‘demand’ pacemakers; and isolating the pacemaker’s battery in a separate compartment to deal with the problem of leaking mercury-zinc batteries.
In 1965, Australian medical device pioneer Noel Gray established Telectronics – Australia’s first manufacturing facility for producing pacemakers that were designed in-house. Telectronics was an innovative developer, achieving some major successes in the early cardiac pacing field, for example, Telectronics’ leads allowed narrowing the pacing pulse to its current nominal of 0.5 milliseconds; encapsulating the pacemaker in titanium instead of epoxy; using a microplasma weld to join the two halves of the pacemaker capsule; creating one of the first rate-responsive ‘demand’ pacemakers; and isolating the pacemaker’s battery in a separate compartment to deal with the problem of leaking mercury-zinc batteries. 


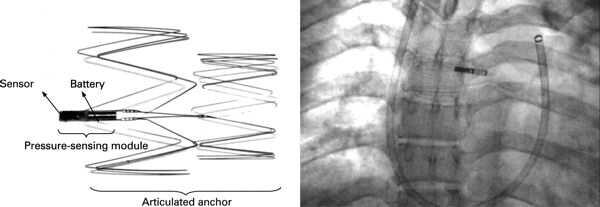
 Sensors for Medicine and Science, Inc. (SMSI) of Germantown, MD was founded in 1997 to develop chemical sensing technologies based on fluorescence sensing.
Sensors for Medicine and Science, Inc. (SMSI) of Germantown, MD was founded in 1997 to develop chemical sensing technologies based on fluorescence sensing.





