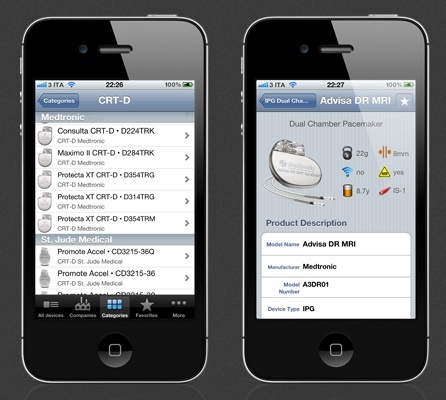
My friend and colleague Dr. Irit Yaniv alerted me to this iPhone app that was just released. It is an implantable pacemaker and defibrillator database that, according to its author, displays up to 70 parameters for each model, includes battery and longevity data, and links directly to product manuals. Continue reading
Category Archives: Heart Failure (CHF)
Implantable devices for the treatment of Congestive Heart Failure (CHF)
Remon Medical’s Implantable Pulmonary Pressure Sensor (1997-2007)
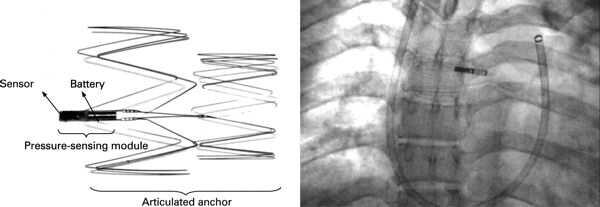
Remon Medical Technologies, Ltd. was founded in 1997 in Caesarea, Israel to develop implantable, wireless pressure sensors.
Remon developed an implantable hemodynamic monitor, which allowed on-demand, non-invasive, leadless self-monitoring of pulmonary artery pressure by the patient at home. ImPressure devices were placed in the pulmonary artery, and transmitted pressure readings to a hand-held monitor. It was hoped that the system would provide early warning of the need for treatment, avoiding hospitalization and deterioration in the patient’s condition. Continue reading
Medtronic’s Chronicle Implantable Hemodynamic Monitor for Heart Failure Monitoring

- Image Credit: Medtronic
The Medtronic Chronicle implantable hemodynamic monitor used a specialized RV lead/sensor. The device was able to monitor and telemeter:
- Systolic and diastolic pressure
- Estimated pulmonary artery diastolic pressure
- RV dp/dt (positive & negative)
- Heart rate & activity
- Core body temperature
- Continuous remote monitoring
- Continue reading
CardioMEMS Miniature Implantable Wireless Sensors

CardioMEMS was founded by Dr. Jay S. Yadav and Dr. Mark G. Allen in Atlanta, GA in 2000 to develop implantable micro-electromechanical sensors to improve the management of severe chronic cardiovascular diseases such as heart failure and aneurysms.
The miniature wireless sensors can be delivered through a catheter. Once in place, they transmit cardiac output, blood pressure and heart rate data that are critical to the management of patients. Due to their small size, durability, and lack of wires and batteries, CardioMEMS’ sensors are designed to be permanently implanted into the cardiovascular system. An external reader is used to interrogate the sensors. Continue reading
BioControl’s CardioFit System for the Treatment of Heart Failure via Vagus Nerve Stimulation
BioControl Medical, Ltd. was founded in 1999 by Yossi Gross, and is based in Yehud, Israel. BioControl’s first devices were developed to treat incontinence due to overactive bladder, stress, and interstitial cystitis. In 2006 American Medical Sytems acquired an exclusive license for the use of the technology in urology, gynecology and other pelvic health applications. Continue reading
EBR System’s Wireless Pacemaker

EBR Systems, Inc., founded in 2003 and headquartered in Sunnyvale, CA, is developing the WiCS® Wireless Cardiac Stimulation technology to eliminate cardiac pacing leads, historically a major source of complications and reliability issues. The startup was spun out of research by founder Debra Echt, a former professor of medicine and a cardiologist at Vanderbilt University. Continue reading
Enopace Biomedical
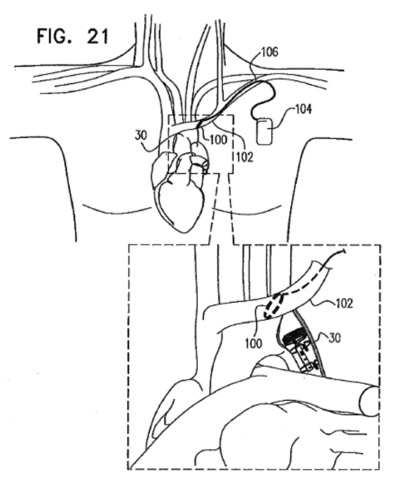
From Patent Application Publication: US 2011/0137370 A1
Enopace Biomedical Ltd. was founded in 2008 by Yossi Gross in Caesarea, Israel.
Very scant details are available besides that it is developing a neuromodulation system to treat patients with congestive heart failure. According to the company, its technology consists of a minimally invasive, implantable neurostimulation device that increases cardiac efficiency by reducing left ventricular workload.
From Enopace’s patent applications, it seems that Enopace’s device consists of a stimulation unit that is placed within the aorta close to the heart, and which is driven wirelessly from a coil that is placed within a vein closeby to the artery. The transmitting coil is in turn driven from an implanted control unit that is implanted subcutaneously. Continue reading
Respicardia’s remedē Implantable Device for the Treatment of Central Sleep Apnea

Image Credit: Respicardia
Respicardia, Inc. (formerly Cardiac Concepts, Inc.) is a privately held medical device company incorporated in 2006. It is located in Minneapolis, Minnesota, USA. It is developing an implantable system intended to improve the outcomes for Heart Failure patients by developing an implantable stimulation therapy to treat Central Sleep Apnea.
The remedē System is an implantable medical device that is being investigated in a clinical research study for the treatment of Central Sleep Apnea in patients with Heart Failure. It consists of three implantable components:
- remedē Pulse Generator comprised of electronic circuitry components and a battery, which are hermetically sealed in a titanium case
- remedē Stimulation Lead that is implanted in a vein close to one of the phrenic nerves
- a sensing lead to detect respiration Continue reading
Angel Medical’s Guardian Implantable Heart Attack Monitor

Photo Credit: PRNewsFoto/Angel Medical Systems
Angel Medical was founded by Dr. Robert E. Fischell and Dr. David R. Fischell. The AngelMed Guardian® system is an implantable cardiac device, designed to detect rapid ST segment shifts that may signify major cardiac events, such as coronary artery occlusions caused by life-threatening vulnerable plaque ruptures. Once an ST shift is detected, the system is designed to alert patients to seek medical care by delivering a series of vibratory, auditory, and visual warnings. The AngelMed Guardian system is intended to address the challenge of patients not recognizing nor responding quickly to heart attacks. Continue reading
AIMD Companies Founded by Yossi Gross
 Yossi Gross (born February 5, 1947) is an Israeli medical device innovator and entrepreneur. His medical-device companies are incubated through Rainbow Medical.
Yossi Gross (born February 5, 1947) is an Israeli medical device innovator and entrepreneur. His medical-device companies are incubated through Rainbow Medical.
Currently, the following companies led by Yossi Gross are focusing on active implantable medical devices:
- BetaStim, developing a new neurostimulation treatment for diabetes
- Brainsgate, developing an implantable device for acute stroke treatment and brain drug-delivery technology
- BioControl Medical, developing neurostimulation devices
- Enopace Biomedical, developing implantable devices for heart failure treatment through stimulation of the aorta
- CerebralRx, a spin-off from Biocontrol developing vagus nerve stimulators for the treatment of epilepsy
AIMD Companies Founded by Dr. Shlomo Ben-Haim

Dr. Shlomo Ben-Haim has held faculty appointments as a Professor of Medicine, Physiology and Biophysics, at both Harvard University and the Technion – Israel Institute of Technology. His formal education is in the fields of nuclear physics, mathematics, biomedical engineering, philosophy, and medicine. Dr. Ben-Haim has published his research in over 100 scientific papers, in peer-reviewed journals. He holds over 180 patents and patent applications.
Currently, two companies led by Dr. Ben-Haim are focusing on active implantable medical devices:
-
Impulse Dynamics, which developed the OPTIMIZER System for the treatment of heart failure
-
MetaCure, which developed the TANTALUS System for the treatment of metabolic syndrome
MTC’s Piezos Used for Wireless Power Transmission in EBR’s Wireless Pacemaker
 Morgan Technical Ceramics (MTC) announced that its Bedford, Ohio site manufactures the piezoelectric ceramic materials used in EBR Systems, Inc.’s innovative new WiCS® Wireless Cardiac Stimulation System. The PZT (lead zirconate titanate) material made by MTC is critical to the efficiency of the WiCS system, which is powered by a battery with a 10-year lifespan.
Morgan Technical Ceramics (MTC) announced that its Bedford, Ohio site manufactures the piezoelectric ceramic materials used in EBR Systems, Inc.’s innovative new WiCS® Wireless Cardiac Stimulation System. The PZT (lead zirconate titanate) material made by MTC is critical to the efficiency of the WiCS system, which is powered by a battery with a 10-year lifespan.
The WiCS technology, which recently began clinical trials, is the first truly wireless pacing device. It was developed to eliminate cardiac pacing leads, historically a major source of complications and reliability issues. Continue reading
Impulse Dynamics’ OPTIMIZER III IPG for Treatment of Heart Failure
 Impulse Dynamics’ OPTIMIZER™ III Implantable Pulse Generator delivers Cardiac Contractility Modulation (CCM), non-excitatory electrical signals during the myocardial absolute refractory period in synchrony with locally sensed electrical activity. CCM signals are intended to treat patients with symptomatic heart failure despite appropriate medical treatment. Continue reading
Impulse Dynamics’ OPTIMIZER™ III Implantable Pulse Generator delivers Cardiac Contractility Modulation (CCM), non-excitatory electrical signals during the myocardial absolute refractory period in synchrony with locally sensed electrical activity. CCM signals are intended to treat patients with symptomatic heart failure despite appropriate medical treatment. Continue reading